Drug Information
Drug (ID: DG00257) and It's Reported Resistant Information
| Name |
Enoxacin
|
||||
|---|---|---|---|---|---|
| Synonyms |
Almitil; Bactidan; Comprecin; Enoram; Enoxacine; Enoxacino; Enoxacinum; Enoxin; Enoxor; Flumark; Penetrex; Enoxacin Sesquihydrate; Enoxacine [French]; Enoxacino [Spanish]; Enoxacinum [Latin]; Faulding Brand of Enoxacin; Pierre Fabre Brand of Enoxacin Sesquihydrate; Rhone Poulenc Rorer Brand of Enoxacin Sesquihydrate; AT 2266; AT2266; CI919; CL23362; E0762; PD 107779; PD107779; AT-2266; Almitil (TN); Bactidan (TN); Bactidron (TN); Comprecin (TN); Enoksetin (TN); Enoxen (TN); Enoxin (TN); Enoxor (TN); Enroxil (TN); Flumark (TN); Gyramid (TN); PD-107779; Penetrex (TN); Rhone-Poulenc Rorer Brand of Enoxacin Sesquihydrate; Sesquihydrate, Enoxacin; Vinone (TN); Enoxacin (USAN/INN); Enoxacin [USAN:BAN:INN:JAN]; 1,8-Naphthyridine-3-carboxylic acid, 6-fluoro-1,4-dihydro-4-oxo-7-piperazinyl; 1-Ethyl-6-fluoro-1,4-dihydro-4-oxo-7-(1-piperazinyl)-1,8-naphthyridine-3-carboxylic acid; 1-Ethyl-6-fluoro-1,4-dihydro-4-oxo-7-[1-piperazinyl]-1,8-naphthyridine-3-carboxylic acid; 1-Ethyl-6-fluoro-4-oxo-7-piperazin-1-yl-1,4-dihydro-[1,8]naphthyridine-3-carboxylic acid; 1-ethyl-6-fluoro-4-oxo-7-(piperazin-1-yl)-1,4-dihydro-1,8-naphthyridine-3-carboxylic acid; 1-ethyl-6-fluoro-4-oxo-7-piperazin-1-yl-1,4-dihydro-1,8-naphthyridine-3-carboxylic acid; 1-ethyl-6-fluoro-4-oxo-7-piperazin-1-yl-1,8-naphthyridine-3-carboxylic acid
Click to Show/Hide
|
||||
| Indication |
In total 1 Indication(s)
|
||||
| Structure |
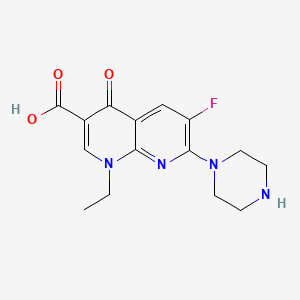
|
||||
| Drug Resistance Disease(s) |
Disease(s) with Clinically Reported Resistance for This Drug
(5 diseases)
[5]
[1]
[6]
[5]
|
||||
| Target | DNA topoisomerase II (TOP2) |
TOP2A_HUMAN
; TOP2B_HUMAN |
[1] | ||
| Click to Show/Hide the Molecular Information and External Link(s) of This Drug | |||||
| Formula |
C15H17FN4O3
|
||||
| IsoSMILES |
CCN1C=C(C(=O)C2=CC(=C(N=C21)N3CCNCC3)F)C(=O)O
|
||||
| InChI |
1S/C15H17FN4O3/c1-2-19-8-10(15(22)23)12(21)9-7-11(16)14(18-13(9)19)20-5-3-17-4-6-20/h7-8,17H,2-6H2,1H3,(H,22,23)
|
||||
| InChIKey |
IDYZIJYBMGIQMJ-UHFFFAOYSA-N
|
||||
| PubChem CID | |||||
| ChEBI ID | |||||
| TTD Drug ID | |||||
| VARIDT ID | |||||
| DrugBank ID | |||||
Type(s) of Resistant Mechanism of This Drug
Drug Resistance Data Categorized by Their Corresponding Diseases
ICD-01: Infectious/parasitic diseases
| Drug Resistance Data Categorized by Their Corresponding Mechanisms | ||||
|
|
||||
| Key Molecule: DNA gyrase subunit A (GYRA) | [2], [3], [4] | |||
| Resistant Disease | Bacterial infection [ICD-11: 1A00-1C4Z] | |||
| Molecule Alteration | Missense mutation | p.S83L |
||
| Experimental Note | Identified from the Human Clinical Data | |||
| In Vitro Model | Escherichia coli strain kL16 | 1425342 | ||
| Escherichia coli strain N-112 | 562 | |||
| Escherichia coli strain N-118 | 562 | |||
| Escherichia coli strain N-119 | 562 | |||
| Escherichia coli strain N-51 | 562 | |||
| Experiment for Molecule Alteration |
Whole genome sequence assay | |||
| Mechanism Description | Quinolones are considered to exert antibacterial activity by inhibiting DNA gyrase (EC 5.99.1.3), which catalyzes topological changes of DNA.DNA gyrase of Escherichia coli consists of subunits A and B, which are the products of the gyrA and gyrB genes, respectively. Mutations in either gene can cause quinolone resistance. | |||
| Key Molecule: DNA gyrase subunit A (GYRA) | [2], [3], [4] | |||
| Resistant Disease | Bacterial infection [ICD-11: 1A00-1C4Z] | |||
| Molecule Alteration | Missense mutation | p.S83W |
||
| Experimental Note | Identified from the Human Clinical Data | |||
| In Vitro Model | Escherichia coli strain kL16 | 1425342 | ||
| Escherichia coli strain P-18 | 562 | |||
| Experiment for Molecule Alteration |
Whole genome sequence assay | |||
| Mechanism Description | Quinolones are considered to exert antibacterial activity by inhibiting DNA gyrase (EC 5.99.1.3), which catalyzes topological changes of DNA.DNA gyrase of Escherichia coli consists of subunits A and B, which are the products of the gyrA and gyrB genes, respectively. Mutations in either gene can cause quinolone resistance. | |||
| Key Molecule: DNA gyrase subunit A (GYRA) | [2], [3], [4] | |||
| Resistant Disease | Bacterial infection [ICD-11: 1A00-1C4Z] | |||
| Molecule Alteration | Missense mutation | p.D87N |
||
| Experimental Note | Identified from the Human Clinical Data | |||
| In Vitro Model | Escherichia coli strain kL16 | 1425342 | ||
| Escherichia coli strain N-113 | 562 | |||
| Experiment for Molecule Alteration |
Whole genome sequence assay | |||
| Mechanism Description | Quinolones are considered to exert antibacterial activity by inhibiting DNA gyrase (EC 5.99.1.3), which catalyzes topological changes of DNA.DNA gyrase of Escherichia coli consists of subunits A and B, which are the products of the gyrA and gyrB genes, respectively. Mutations in either gene can cause quinolone resistance. | |||
| Key Molecule: DNA gyrase subunit A (GYRA) | [2], [3], [4] | |||
| Resistant Disease | Bacterial infection [ICD-11: 1A00-1C4Z] | |||
| Molecule Alteration | Missense mutation | p.G81C |
||
| Experimental Note | Identified from the Human Clinical Data | |||
| In Vitro Model | Escherichia coli strain kL16 | 1425342 | ||
| Escherichia coli strain N-97 | 562 | |||
| Experiment for Molecule Alteration |
Whole genome sequence assay | |||
| Mechanism Description | Quinolones are considered to exert antibacterial activity by inhibiting DNA gyrase (EC 5.99.1.3), which catalyzes topological changes of DNA.DNA gyrase of Escherichia coli consists of subunits A and B, which are the products of the gyrA and gyrB genes, respectively. Mutations in either gene can cause quinolone resistance. | |||
| Key Molecule: DNA gyrase subunit A (GYRA) | [2], [3], [4] | |||
| Resistant Disease | Bacterial infection [ICD-11: 1A00-1C4Z] | |||
| Molecule Alteration | Missense mutation | p.A84P |
||
| Experimental Note | Identified from the Human Clinical Data | |||
| In Vitro Model | Escherichia coli strain kL16 | 1425342 | ||
| Escherichia coli strain P-5 | 562 | |||
| Experiment for Molecule Alteration |
Whole genome sequence assay | |||
| Mechanism Description | Quinolones are considered to exert antibacterial activity by inhibiting DNA gyrase (EC 5.99.1.3), which catalyzes topological changes of DNA.DNA gyrase of Escherichia coli consists of subunits A and B, which are the products of the gyrA and gyrB genes, respectively. Mutations in either gene can cause quinolone resistance. | |||
| Key Molecule: DNA gyrase subunit A (GYRA) | [2], [3], [4] | |||
| Resistant Disease | Bacterial infection [ICD-11: 1A00-1C4Z] | |||
| Molecule Alteration | Missense mutation | p.A67S |
||
| Experimental Note | Identified from the Human Clinical Data | |||
| In Vitro Model | Escherichia coli strain kL16 | 1425342 | ||
| Escherichia coli strain P-10 | 562 | |||
| Experiment for Molecule Alteration |
Whole genome sequence assay | |||
| Mechanism Description | Quinolones are considered to exert antibacterial activity by inhibiting DNA gyrase (EC 5.99.1.3), which catalyzes topological changes of DNA.DNA gyrase of Escherichia coli consists of subunits A and B, which are the products of the gyrA and gyrB genes, respectively. Mutations in either gene can cause quinolone resistance. | |||
| Key Molecule: DNA gyrase subunit A (GYRA) | [2], [3], [4] | |||
| Resistant Disease | Bacterial infection [ICD-11: 1A00-1C4Z] | |||
| Molecule Alteration | Missense mutation | p.Q106H |
||
| Experimental Note | Identified from the Human Clinical Data | |||
| In Vitro Model | Escherichia coli strain kL16 | 1425342 | ||
| Escherichia coli strain N-89 | 562 | |||
| Experiment for Molecule Alteration |
Whole genome sequence assay | |||
| Mechanism Description | Quinolones are considered to exert antibacterial activity by inhibiting DNA gyrase (EC 5.99.1.3), which catalyzes topological changes of DNA.DNA gyrase of Escherichia coli consists of subunits A and B, which are the products of the gyrA and gyrB genes, respectively. Mutations in either gene can cause quinolone resistance. | |||
| Drug Resistance Data Categorized by Their Corresponding Mechanisms | ||||
|
|
||||
| Key Molecule: Quinolone resistance protein NorA (NORA) | [5] | |||
| Resistant Disease | Escherichia coli infection [ICD-11: 1A03.0] | |||
| Molecule Alteration | Expression | Acquired |
||
| Experimental Note | Identified from the Human Clinical Data | |||
| In Vitro Model | Escherichia coli HB101 | 634468 | ||
| Staphylococcus aureus strain SA113 | 1280 | |||
| Experiment for Molecule Alteration |
Dideoxy chain-termination method assay | |||
| Mechanism Description | The norA gene cloned from chromosomal DNA of quinolone-resistant Staphylococcus aureus Tk2566 conferred relatively high resistance to hydrophilic quinolones such as norfloxacin, enoxacin, ofloxacin, and ciprofloxacin, but only low or no resistance at all to hydrophobic ones such as nalidixic acid, oxolinic acid, and sparfloxacin in S. aureus and Escherichia coli. Escherichia coli strains containing one of the plasmids carrying the norA gene (pTUS1, pTUS180, pTUS829, and pTUS206) were 8 to 64 times more resistant to the hydrophilic quinolones than the parent quinolone-susceptible strain. | |||
| Drug Resistance Data Categorized by Their Corresponding Mechanisms | ||||
|
|
||||
| Key Molecule: DNA topoisomerase 4 subunit B (PARE) | [1] | |||
| Resistant Disease | Leprosy [ICD-11: 1B20.0] | |||
| Molecule Alteration | Missense mutation | p.D464N |
||
| Experimental Note | Identified from the Human Clinical Data | |||
| In Vitro Model | Escherichia coli BL21 (DE3) | 469008 | ||
| Escherichia coli Rosetta-gami 2 | 562 | |||
| Escherichia coli TOP-10 | 83333 | |||
| Mycobacterium leprae Thai-53 | 1769 | |||
| Experiment for Molecule Alteration |
Whole genome sequence assay; Allelic frequency measurement assay | |||
| Experiment for Drug Resistance |
DNA supercoiling assay; DNA cleavage assay | |||
| Mechanism Description | FQs are known to interact with both A and B subunits of DNA gyrase and inhibit supercoiling activity of this enzyme.The FQ-inhibited supercoiling assay and FQ-induced cleavage assay demonstrated the important roles of these amino acid substitutions in reduced sensitivity to FQ with marked influence by amino acid substitution, especially at position 502. | |||
| Key Molecule: DNA topoisomerase 4 subunit B (PARE) | [1] | |||
| Resistant Disease | Leprosy [ICD-11: 1B20.0] | |||
| Molecule Alteration | Missense mutation | p.N502D |
||
| Experimental Note | Identified from the Human Clinical Data | |||
| In Vitro Model | Escherichia coli BL21 (DE3) | 469008 | ||
| Escherichia coli Rosetta-gami 2 | 562 | |||
| Escherichia coli TOP-10 | 83333 | |||
| Mycobacterium leprae Thai-53 | 1769 | |||
| Experiment for Molecule Alteration |
Whole genome sequence assay; Allelic frequency measurement assay | |||
| Experiment for Drug Resistance |
DNA supercoiling assay; DNA cleavage assay | |||
| Mechanism Description | FQs are known to interact with both A and B subunits of DNA gyrase and inhibit supercoiling activity of this enzyme.The FQ-inhibited supercoiling assay and FQ-induced cleavage assay demonstrated the important roles of these amino acid substitutions in reduced sensitivity to FQ with marked influence by amino acid substitution, especially at position 502. | |||
| Key Molecule: DNA topoisomerase 4 subunit B (PARE) | [1] | |||
| Resistant Disease | Leprosy [ICD-11: 1B20.0] | |||
| Molecule Alteration | Missense mutation | p.E504V |
||
| Experimental Note | Identified from the Human Clinical Data | |||
| In Vitro Model | Escherichia coli BL21 (DE3) | 469008 | ||
| Escherichia coli Rosetta-gami 2 | 562 | |||
| Escherichia coli TOP-10 | 83333 | |||
| Mycobacterium leprae Thai-53 | 1769 | |||
| Experiment for Molecule Alteration |
Whole genome sequence assay; Allelic frequency measurement assay | |||
| Experiment for Drug Resistance |
DNA supercoiling assay; DNA cleavage assay | |||
| Mechanism Description | FQs are known to interact with both A and B subunits of DNA gyrase and inhibit supercoiling activity of this enzyme.The FQ-inhibited supercoiling assay and FQ-induced cleavage assay demonstrated the important roles of these amino acid substitutions in reduced sensitivity to FQ with marked influence by amino acid substitution, especially at position 502. | |||
| Drug Resistance Data Categorized by Their Corresponding Mechanisms | ||||
|
|
||||
| Key Molecule: Quinolone resistance protein NorA (NORA) | [5] | |||
| Resistant Disease | Staphylococcus aureus infection [ICD-11: 1B54.0] | |||
| Molecule Alteration | Expression | Inherence |
||
| Experimental Note | Identified from the Human Clinical Data | |||
| In Vitro Model | Escherichia coli HB101 | 634468 | ||
| Staphylococcus aureus strain SA113 | 1280 | |||
| Experiment for Molecule Alteration |
Dideoxy chain-termination method assay | |||
| Mechanism Description | The norA gene cloned from chromosomal DNA of quinolone-resistant Staphylococcus aureus Tk2566 conferred relatively high resistance to hydrophilic quinolones such as norfloxacin, enoxacin, ofloxacin, and ciprofloxacin, but only low or no resistance at all to hydrophobic ones such as nalidixic acid, oxolinic acid, and sparfloxacin in S. aureus and Escherichia coli. | |||
| Key Molecule: Quinolone resistance protein NorA (NORA) | [5] | |||
| Resistant Disease | Staphylococcus aureus infection [ICD-11: 1B54.0] | |||
| Molecule Alteration | Expression | Acquired |
||
| Experimental Note | Identified from the Human Clinical Data | |||
| In Vitro Model | Escherichia coli HB101 | 634468 | ||
| Staphylococcus aureus strain SA113 | 1280 | |||
| Experiment for Molecule Alteration |
Dideoxy chain-termination method assay | |||
| Mechanism Description | The norA gene cloned from chromosomal DNA of quinolone-resistant Staphylococcus aureus Tk2566 conferred relatively high resistance to hydrophilic quinolones such as norfloxacin, enoxacin, ofloxacin, and ciprofloxacin, but only low or no resistance at all to hydrophobic ones such as nalidixic acid, oxolinic acid, and sparfloxacin in S. aureus and Escherichia coli. S. aureus SA113 (pTUS20) harboring a plasmid carrying the staphylococcal norA gene was 16 to 64 times more resistant to relatively hydrophilic quinolones. | |||
ICD-12: Respiratory system diseases
| Drug Resistance Data Categorized by Their Corresponding Mechanisms | ||||
|
|
||||
| Key Molecule: DNA gyrase subunit A (GYRA) | [6] | |||
| Resistant Disease | Respiratory trac infection [ICD-11: CA45.0] | |||
| Molecule Alteration | Missense mutation | p.G75S |
||
| Experimental Note | Identified from the Human Clinical Data | |||
| In Vitro Model | Staphylococcus aureus ATCC 29213 | 1280 | ||
| Staphylococcus aureus isolates | 1280 | |||
| Experiment for Drug Resistance |
Broth microdilution method assay | |||
| Mechanism Description | Quinolone/fluoroquinolone resistance is most likely due to mutations in the genes gyrA and parC encoding DNA gyrase and topoisomerase IV. | |||
| Key Molecule: DNA gyrase subunit A (GYRA) | [6] | |||
| Resistant Disease | Respiratory trac infection [ICD-11: CA45.0] | |||
| Molecule Alteration | Missense mutation | p.S83R |
||
| Experimental Note | Identified from the Human Clinical Data | |||
| In Vitro Model | Staphylococcus aureus ATCC 29213 | 1280 | ||
| Staphylococcus aureus isolates | 1280 | |||
| Experiment for Drug Resistance |
Broth microdilution method assay | |||
| Mechanism Description | Quinolone/fluoroquinolone resistance is most likely due to mutations in the genes gyrA and parC encoding DNA gyrase and topoisomerase IV. | |||
References
If you find any error in data or bug in web service, please kindly report it to Dr. Sun and Dr. Yu.
