Drug Information
Drug (ID: DG00019) and It's Reported Resistant Information
| Name |
Fexofenadine
|
||||
|---|---|---|---|---|---|
| Synonyms |
Carboxyterfenadine; Fexofendine; Terfenadine acid metabolite; Terfenadine carboxylate; F 9427; MDL 16455; Allegra (TN); Fastofen (TN); Fexofenadine (INN); Fexofenadine [INN:BAN]; Telfast (TN); Terfenadine-COOH; Terfenidine carboxylate, MDL 16455; Tilfur (TN); 2-[4-(1-hydroxy-4-{4-[hydroxy(diphenyl)methyl]piperidin-1-yl}butyl)phenyl]-2-methylpropanoic acid; 2-[4-[1-hydroxy-4-[4-[hydroxy(diphenyl)methyl]piperidin-1-yl]butyl]phenyl]-2-methylpropanoic acid; 4-(1-Hydroxy-4-(4-(hydroxydiphenylmethyl)-1-piperidinyl)butyl)-alpha,alpha-dimethylbenzeneacetic acid
Click to Show/Hide
|
||||
| Indication |
In total 1 Indication(s)
|
||||
| Structure |
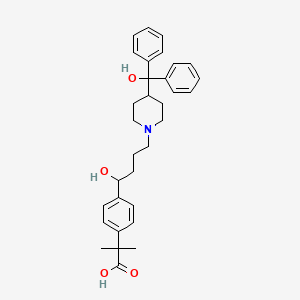
|
||||
| Target | Histamine H1 receptor (H1R) | HRH1_HUMAN | [1] | ||
| Click to Show/Hide the Molecular Information and External Link(s) of This Drug | |||||
| Formula |
C32H39NO4
|
||||
| IsoSMILES |
CC(C)(C1=CC=C(C=C1)C(CCCN2CCC(CC2)C(C3=CC=CC=C3)(C4=CC=CC=C4)O)O)C(=O)O
|
||||
| InChI |
1S/C32H39NO4/c1-31(2,30(35)36)25-17-15-24(16-18-25)29(34)14-9-21-33-22-19-28(20-23-33)32(37,26-10-5-3-6-11-26)27-12-7-4-8-13-27/h3-8,10-13,15-18,28-29,34,37H,9,14,19-23H2,1-2H3,(H,35,36)
|
||||
| InChIKey |
RWTNPBWLLIMQHL-UHFFFAOYSA-N
|
||||
| PubChem CID | |||||
| ChEBI ID | |||||
| TTD Drug ID | |||||
| VARIDT ID | |||||
| DrugBank ID | |||||
Type(s) of Resistant Mechanism of This Drug
Drug Resistance Data Categorized by Their Corresponding Diseases
ICD-12: Respiratory system diseases
| Drug Sensitivity Data Categorized by Their Corresponding Mechanisms | ||||
|
|
||||
| Key Molecule: ATP-binding cassette sub-family B5 (ABCB5) | [1] | |||
| Sensitive Disease | Seasonal allergic rhinitis [ICD-11: CA08.0] | |||
| Molecule Alteration | Expression | Down-regulation |
||
| Experimental Note | Identified from the Human Clinical Data | |||
| Experiment for Molecule Alteration |
qPCR | |||
| Experiment for Drug Resistance |
Ussing chamber system assay | |||
| Mechanism Description | Digoxin and fexofenadine (each 5 uM) were selected as P-gp substrates, and sulfasalazine and rosuvastatin (each 5 uM) were selected as BCRP substrates to evaluate the efflux transport mediated by P-gp and BCRP. PSC833 (15 uM) and ko143 (15 uM) were used as typical inhibitors of P-gp and BCRP, respectively. Serosal-to-mucosal transport of all the tested P-gp and BCRP substrate drugs was significantly decreased or tended to decrease in the presence of P-gp/BCRP inhibitor cocktail. | |||
References
If you find any error in data or bug in web service, please kindly report it to Dr. Sun and Dr. Yu.
