Drug Information
Drug (ID: DG00269) and It's Reported Resistant Information
| Name |
Rilpivirine
|
||||
|---|---|---|---|---|---|
| Synonyms |
500287-72-9; TMC278; Edurant; TMC 278; UNII-FI96A8X663; R278474; TMC-278; 4-{[4-({4-[(E)-2-Cyanoethenyl]-2,6-Dimethylphenyl}amino)pyrimidin-2-Yl]amino}benzonitrile; CHEBI:68606; FI96A8X663; R 278474; 4-{[4-({4-[(E)-2-cyanovinyl]-2,6-dimethylphenyl}amino)pyrimidin-2-yl]amino}benzonitrile; (E)-4-((4-((4-(2-cyanovinyl)-2,6-dimethylphenyl)amino)pyrimidin-2-yl)amino)benzonitrile; (E)-4-(4-(4-(2-cyanovinyl)-2,6-dimethylphenylamino)pyrimidin-2-ylamino)benzonitrile; W-202888; RPV; Mu O-conotoxin
Click to Show/Hide
|
||||
| Indication |
In total 2 Indication(s)
|
||||
| Structure |
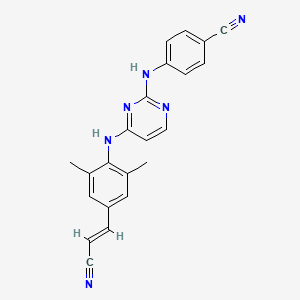
|
||||
| Drug Resistance Disease(s) |
Disease(s) with Clinically Reported Resistance for This Drug
(1 diseases)
[2]
Disease(s) with Resistance Information Validated by in-vivo Model for This Drug
(1 diseases)
[1]
|
||||
| Target | Human immunodeficiency virus Reverse transcriptase (HIV RT) | POL_HV1B1 | [1] | ||
| Voltage-gated sodium channel alpha Nav1.8 (SCN10A) | SCNAA_HUMAN | [1] | |||
| Click to Show/Hide the Molecular Information and External Link(s) of This Drug | |||||
| Formula |
C22H18N6
|
||||
| IsoSMILES |
CC1=CC(=CC(=C1NC2=NC(=NC=C2)NC3=CC=C(C=C3)C#N)C)/C=C/C#N
|
||||
| InChI |
1S/C22H18N6/c1-15-12-18(4-3-10-23)13-16(2)21(15)27-20-9-11-25-22(28-20)26-19-7-5-17(14-24)6-8-19/h3-9,11-13H,1-2H3,(H2,25,26,27,28)/b4-3+
|
||||
| InChIKey |
YIBOMRUWOWDFLG-ONEGZZNKSA-N
|
||||
| PubChem CID | |||||
| ChEBI ID | |||||
| TTD Drug ID | |||||
| DrugBank ID | |||||
Type(s) of Resistant Mechanism of This Drug
Drug Resistance Data Categorized by Their Corresponding Diseases
ICD-01: Infectious/parasitic diseases
| Drug Resistance Data Categorized by Their Corresponding Mechanisms | |||||||||||||
|
|
|||||||||||||
| Key Molecule: HIV1 Reverse transcriptase (HIV1 RT) | [3] | ||||||||||||
| Resistant Disease | Human immunodeficiency virus infection [ICD-11: 1C62.0] | ||||||||||||
| Molecule Alteration | Missense mutation | p.S48T+p.V90VI+p.K101E+p.Y115YF+p.Y181YC+p.M184I+p.R211N+p.K219KE+p.V245N+p.L283I+p.V292I+p.I293V |
|||||||||||
| Wild Type Structure | Method: X-ray diffraction | Resolution: 2.09 Å | |||||||||||
| Mutant Type Structure | Method: AlphaFold | Average pLDDT: 86.992 |

|
||||||||||
| Download The Information of Sequence | Download The Structure File | ||||||||||||
-
P
P
I
I
S
S
P
P
I
I
E
E
T
T
V
V
P
P
10
|
V
V
K
K
L
L
K
K
P
P
G
G
M
M
D
D
G
G
P
P
20
|
K
K
V
V
K
K
Q
Q
W
W
P
P
L
L
T
T
E
E
E
E
30
|
K
K
I
I
K
K
A
A
L
L
V
V
E
E
I
I
C
C
T
T
40
|
E
E
M
M
E
E
K
K
E
E
G
G
K
K
I
I
S
T
K
K
50
|
I
I
G
G
P
P
E
E
N
N
P
P
Y
Y
N
N
T
T
P
P
60
|
V
V
F
F
A
A
I
I
K
K
K
K
K
K
D
D
S
S
T
T
70
|
K
K
W
W
R
R
K
K
L
L
V
V
D
D
F
F
R
R
E
E
80
|
L
L
N
N
K
K
R
R
T
T
Q
Q
D
D
F
F
W
W
E
E
90
|
V
I
Q
Q
L
L
G
G
I
I
P
P
H
H
P
P
A
A
G
G
100
|
L
L
K
E
K
K
K
K
K
K
S
S
V
V
T
T
V
V
L
L
110
|
D
D
V
V
G
G
D
D
A
A
Y
F
F
F
S
S
V
V
P
P
120
|
L
L
D
D
E
E
D
D
F
F
R
R
K
K
Y
Y
T
T
A
A
130
|
F
F
T
T
I
I
P
P
S
S
I
I
N
N
N
N
E
E
T
T
140
|
P
P
G
G
I
I
R
R
Y
Y
Q
Q
Y
Y
N
N
V
V
L
L
150
|
P
P
Q
Q
G
G
W
W
K
K
G
G
S
S
P
P
A
A
I
I
160
|
F
F
Q
Q
S
S
S
S
M
M
T
T
K
K
I
I
L
L
E
E
170
|
P
P
F
F
R
R
K
K
Q
Q
N
N
P
P
D
D
I
I
V
V
180
|
I
I
Y
C
Q
Q
Y
Y
M
I
D
D
D
D
L
L
Y
Y
V
V
190
|
G
G
S
S
D
D
L
L
E
E
I
I
G
G
Q
Q
H
H
R
R
200
|
T
T
K
K
I
I
E
E
E
E
L
L
R
R
Q
Q
H
H
L
L
210
|
L
L
R
N
W
W
G
G
L
L
T
T
T
T
P
P
D
D
K
E
220
|
K
K
H
H
Q
Q
K
K
E
E
P
P
P
P
F
F
L
L
W
W
230
|
M
M
G
G
Y
Y
E
E
L
L
H
H
P
P
D
D
K
K
W
W
240
|
T
T
V
V
Q
Q
P
P
I
I
V
N
L
L
P
P
E
E
K
K
250
|
D
D
S
S
W
W
T
T
V
V
N
N
D
D
I
I
Q
Q
K
K
260
|
L
L
V
V
G
G
K
K
L
L
N
N
W
W
A
A
S
S
Q
Q
270
|
I
I
Y
Y
P
P
G
G
I
I
K
K
V
V
R
R
Q
Q
L
L
280
|
C
C
K
K
L
L
L
I
R
R
G
G
T
T
K
K
A
A
L
L
290
|
T
T
E
E
V
I
I
V
P
P
L
L
T
T
E
E
E
E
A
A
300
|
E
E
L
L
E
E
L
L
A
A
E
E
N
N
R
R
E
E
I
I
310
|
L
L
K
K
E
E
P
P
V
V
H
H
G
G
V
V
Y
Y
Y
Y
320
|
D
D
P
P
S
S
K
K
D
D
L
L
I
I
A
A
E
E
I
I
330
|
Q
Q
K
K
Q
Q
G
G
Q
Q
G
G
Q
Q
W
W
T
T
Y
Y
340
|
Q
Q
I
I
Y
Y
Q
Q
E
E
P
P
F
F
K
K
N
N
L
L
350
|
K
K
T
T
G
G
K
K
Y
Y
A
A
R
R
M
M
R
R
G
G
360
|
A
A
H
H
T
T
N
N
D
D
V
V
K
K
Q
Q
L
L
T
T
370
|
E
E
A
A
V
V
Q
Q
K
K
I
I
T
T
T
T
E
E
S
S
380
|
I
I
V
V
I
I
W
W
G
G
K
K
T
T
P
P
K
K
F
F
390
|
K
K
L
L
P
P
I
I
Q
Q
K
K
E
E
T
T
W
W
E
E
400
|
T
T
W
W
W
W
T
T
E
E
Y
Y
W
W
Q
Q
A
A
T
T
410
|
W
W
I
I
P
P
E
E
W
W
E
E
F
F
V
V
N
N
T
T
420
|
P
P
P
P
L
L
V
V
K
K
L
L
W
W
Y
Y
Q
Q
L
L
430
|
E
E
K
K
E
E
P
P
I
I
V
V
G
G
A
A
E
E
T
T
440
|
F
F
Y
Y
V
V
D
D
G
G
A
A
A
A
N
N
R
R
E
E
450
|
T
T
K
K
L
L
G
G
K
K
A
A
G
G
Y
Y
V
V
T
T
460
|
N
N
R
R
G
G
R
R
Q
Q
K
K
V
V
V
V
T
T
L
L
470
|
T
T
D
D
T
T
T
T
N
N
Q
Q
K
K
T
T
E
E
L
L
480
|
Q
Q
A
A
I
I
Y
Y
L
L
A
A
L
L
Q
Q
D
D
S
S
490
|
G
G
L
L
E
E
V
V
N
N
I
I
V
V
T
T
D
D
S
S
500
|
Q
Q
Y
Y
A
A
L
L
G
G
I
I
I
I
Q
Q
A
A
Q
Q
510
|
P
P
D
D
Q
Q
S
S
E
E
S
S
E
E
L
L
V
V
N
N
520
|
Q
Q
I
I
I
I
E
E
Q
Q
L
L
I
I
K
K
K
K
E
E
530
|
K
K
V
V
Y
Y
L
L
A
A
W
W
V
V
P
P
A
A
H
H
540
|
K
K
G
G
I
I
G
G
G
G
N
N
E
E
Q
Q
V
V
D
D
550
|
K
K
L
L
V
V
S
S
A
A
G
G
I
I
R
R
K
K
V
V
560
|
L
L
|
|||||||||||||
| Experimental Note | Identified from the Human Clinical Data | ||||||||||||
| Experiment for Molecule Alteration |
GeneSeq assay; PhenoSense GT assay | ||||||||||||
| Experiment for Drug Resistance |
Viral load assay | ||||||||||||
| Mechanism Description | Amino acid changes in IN may contribute to drug resistance or sensitivity. | ||||||||||||
| Key Molecule: HIV1 Reverse transcriptase (HIV1 RT) | [3] | ||||||||||||
| Resistant Disease | Human immunodeficiency virus infection [ICD-11: 1C62.0] | ||||||||||||
| Molecule Alteration | Missense mutation | p.V35I+p.L74V+p.L100I+p.K103N+p.M184V+p.Q207A+p.R211K+p.P225H+p.I293V |
|||||||||||
| Wild Type Structure | Method: X-ray diffraction | Resolution: 2.09 Å | |||||||||||
| Mutant Type Structure | Method: AlphaFold | Average pLDDT: 86.928 |

|
||||||||||
| Download The Information of Sequence | Download The Structure File | ||||||||||||
-
P
P
I
I
S
S
P
P
I
I
E
E
T
T
V
V
P
P
10
|
V
V
K
K
L
L
K
K
P
P
G
G
M
M
D
D
G
G
P
P
20
|
K
K
V
V
K
K
Q
Q
W
W
P
P
L
L
T
T
E
E
E
E
30
|
K
K
I
I
K
K
A
A
L
L
V
I
E
E
I
I
C
C
T
T
40
|
E
E
M
M
E
E
K
K
E
E
G
G
K
K
I
I
S
S
K
K
50
|
I
I
G
G
P
P
E
E
N
N
P
P
Y
Y
N
N
T
T
P
P
60
|
V
V
F
F
A
A
I
I
K
K
K
K
K
K
D
D
S
S
T
T
70
|
K
K
W
W
R
R
K
K
L
V
V
V
D
D
F
F
R
R
E
E
80
|
L
L
N
N
K
K
R
R
T
T
Q
Q
D
D
F
F
W
W
E
E
90
|
V
V
Q
Q
L
L
G
G
I
I
P
P
H
H
P
P
A
A
G
G
100
|
L
I
K
K
K
K
K
N
K
K
S
S
V
V
T
T
V
V
L
L
110
|
D
D
V
V
G
G
D
D
A
A
Y
Y
F
F
S
S
V
V
P
P
120
|
L
L
D
D
E
E
D
D
F
F
R
R
K
K
Y
Y
T
T
A
A
130
|
F
F
T
T
I
I
P
P
S
S
I
I
N
N
N
N
E
E
T
T
140
|
P
P
G
G
I
I
R
R
Y
Y
Q
Q
Y
Y
N
N
V
V
L
L
150
|
P
P
Q
Q
G
G
W
W
K
K
G
G
S
S
P
P
A
A
I
I
160
|
F
F
Q
Q
S
S
S
S
M
M
T
T
K
K
I
I
L
L
E
E
170
|
P
P
F
F
R
R
K
K
Q
Q
N
N
P
P
D
D
I
I
V
V
180
|
I
I
Y
Y
Q
Q
Y
Y
M
V
D
D
D
D
L
L
Y
Y
V
V
190
|
G
G
S
S
D
D
L
L
E
E
I
I
G
G
Q
Q
H
H
R
R
200
|
T
T
K
K
I
I
E
E
E
E
L
L
R
R
Q
A
H
H
L
L
210
|
L
L
R
K
W
W
G
G
L
L
T
T
T
T
P
P
D
D
K
K
220
|
K
K
H
H
Q
Q
K
K
E
E
P
H
P
P
F
F
L
L
W
W
230
|
M
M
G
G
Y
Y
E
E
L
L
H
H
P
P
D
D
K
K
W
W
240
|
T
T
V
V
Q
Q
P
P
I
I
V
V
L
L
P
P
E
E
K
K
250
|
D
D
S
S
W
W
T
T
V
V
N
N
D
D
I
I
Q
Q
K
K
260
|
L
L
V
V
G
G
K
K
L
L
N
N
W
W
A
A
S
S
Q
Q
270
|
I
I
Y
Y
P
P
G
G
I
I
K
K
V
V
R
R
Q
Q
L
L
280
|
C
C
K
K
L
L
L
L
R
R
G
G
T
T
K
K
A
A
L
L
290
|
T
T
E
E
V
V
I
V
P
P
L
L
T
T
E
E
E
E
A
A
300
|
E
E
L
L
E
E
L
L
A
A
E
E
N
N
R
R
E
E
I
I
310
|
L
L
K
K
E
E
P
P
V
V
H
H
G
G
V
V
Y
Y
Y
Y
320
|
D
D
P
P
S
S
K
K
D
D
L
L
I
I
A
A
E
E
I
I
330
|
Q
Q
K
K
Q
Q
G
G
Q
Q
G
G
Q
Q
W
W
T
T
Y
Y
340
|
Q
Q
I
I
Y
Y
Q
Q
E
E
P
P
F
F
K
K
N
N
L
L
350
|
K
K
T
T
G
G
K
K
Y
Y
A
A
R
R
M
M
R
R
G
G
360
|
A
A
H
H
T
T
N
N
D
D
V
V
K
K
Q
Q
L
L
T
T
370
|
E
E
A
A
V
V
Q
Q
K
K
I
I
T
T
T
T
E
E
S
S
380
|
I
I
V
V
I
I
W
W
G
G
K
K
T
T
P
P
K
K
F
F
390
|
K
K
L
L
P
P
I
I
Q
Q
K
K
E
E
T
T
W
W
E
E
400
|
T
T
W
W
W
W
T
T
E
E
Y
Y
W
W
Q
Q
A
A
T
T
410
|
W
W
I
I
P
P
E
E
W
W
E
E
F
F
V
V
N
N
T
T
420
|
P
P
P
P
L
L
V
V
K
K
L
L
W
W
Y
Y
Q
Q
L
L
430
|
E
E
K
K
E
E
P
P
I
I
V
V
G
G
A
A
E
E
T
T
440
|
F
F
Y
Y
V
V
D
D
G
G
A
A
A
A
N
N
R
R
E
E
450
|
T
T
K
K
L
L
G
G
K
K
A
A
G
G
Y
Y
V
V
T
T
460
|
N
N
R
R
G
G
R
R
Q
Q
K
K
V
V
V
V
T
T
L
L
470
|
T
T
D
D
T
T
T
T
N
N
Q
Q
K
K
T
T
E
E
L
L
480
|
Q
Q
A
A
I
I
Y
Y
L
L
A
A
L
L
Q
Q
D
D
S
S
490
|
G
G
L
L
E
E
V
V
N
N
I
I
V
V
T
T
D
D
S
S
500
|
Q
Q
Y
Y
A
A
L
L
G
G
I
I
I
I
Q
Q
A
A
Q
Q
510
|
P
P
D
D
Q
Q
S
S
E
E
S
S
E
E
L
L
V
V
N
N
520
|
Q
Q
I
I
I
I
E
E
Q
Q
L
L
I
I
K
K
K
K
E
E
530
|
K
K
V
V
Y
Y
L
L
A
A
W
W
V
V
P
P
A
A
H
H
540
|
K
K
G
G
I
I
G
G
G
G
N
N
E
E
Q
Q
V
V
D
D
550
|
K
K
L
L
V
V
S
S
A
A
G
G
I
I
R
R
K
K
V
V
560
|
L
L
|
|||||||||||||
| Experimental Note | Identified from the Human Clinical Data | ||||||||||||
| Experiment for Molecule Alteration |
GeneSeq assay; PhenoSense GT assay | ||||||||||||
| Experiment for Drug Resistance |
Viral load assay | ||||||||||||
| Mechanism Description | Amino acid changes in IN may contribute to drug resistance or sensitivity. | ||||||||||||
| Key Molecule: HIV1 Reverse transcriptase (HIV1 RT) | [2] | ||||||||||||
| Resistant Disease | Human immunodeficiency virus infection [ICD-11: 1C62.0] | ||||||||||||
| Molecule Alteration | Missense mutation | p.K20R+p.V60I+p.K103N+p.D123E+p.I142V+p.Y181C+p.G196E+p.T200A+p.R211K+p.K219E+p.I293IM |
|||||||||||
| Wild Type Structure | Method: X-ray diffraction | Resolution: 2.09 Å | |||||||||||
| Mutant Type Structure | Method: AlphaFold | Average pLDDT: 86.998 |

|
||||||||||
| Download The Information of Sequence | Download The Structure File | ||||||||||||
-
P
P
I
I
S
S
P
P
I
I
E
E
T
T
V
V
P
P
10
|
V
V
K
K
L
L
K
K
P
P
G
G
M
M
D
D
G
G
P
P
20
|
K
R
V
V
K
K
Q
Q
W
W
P
P
L
L
T
T
E
E
E
E
30
|
K
K
I
I
K
K
A
A
L
L
V
V
E
E
I
I
C
C
T
T
40
|
E
E
M
M
E
E
K
K
E
E
G
G
K
K
I
I
S
S
K
K
50
|
I
I
G
G
P
P
E
E
N
N
P
P
Y
Y
N
N
T
T
P
P
60
|
V
I
F
F
A
A
I
I
K
K
K
K
K
K
D
D
S
S
T
T
70
|
K
K
W
W
R
R
K
K
L
L
V
V
D
D
F
F
R
R
E
E
80
|
L
L
N
N
K
K
R
R
T
T
Q
Q
D
D
F
F
W
W
E
E
90
|
V
V
Q
Q
L
L
G
G
I
I
P
P
H
H
P
P
A
A
G
G
100
|
L
L
K
K
K
K
K
N
K
K
S
S
V
V
T
T
V
V
L
L
110
|
D
D
V
V
G
G
D
D
A
A
Y
Y
F
F
S
S
V
V
P
P
120
|
L
L
D
D
E
E
D
E
F
F
R
R
K
K
Y
Y
T
T
A
A
130
|
F
F
T
T
I
I
P
P
S
S
I
I
N
N
N
N
E
E
T
T
140
|
P
P
G
G
I
V
R
R
Y
Y
Q
Q
Y
Y
N
N
V
V
L
L
150
|
P
P
Q
Q
G
G
W
W
K
K
G
G
S
S
P
P
A
A
I
I
160
|
F
F
Q
Q
S
S
S
S
M
M
T
T
K
K
I
I
L
L
E
E
170
|
P
P
F
F
R
R
K
K
Q
Q
N
N
P
P
D
D
I
I
V
V
180
|
I
I
Y
C
Q
Q
Y
Y
M
M
D
D
D
D
L
L
Y
Y
V
V
190
|
G
G
S
S
D
D
L
L
E
E
I
I
G
E
Q
Q
H
H
R
R
200
|
T
A
K
K
I
I
E
E
E
E
L
L
R
R
Q
Q
H
H
L
L
210
|
L
L
R
K
W
W
G
G
L
L
T
T
T
T
P
P
D
D
K
E
220
|
K
K
H
H
Q
Q
K
K
E
E
P
P
P
P
F
F
L
L
W
W
230
|
M
M
G
G
Y
Y
E
E
L
L
H
H
P
P
D
D
K
K
W
W
240
|
T
T
V
V
Q
Q
P
P
I
I
V
V
L
L
P
P
E
E
K
K
250
|
D
D
S
S
W
W
T
T
V
V
N
N
D
D
I
I
Q
Q
K
K
260
|
L
L
V
V
G
G
K
K
L
L
N
N
W
W
A
A
S
S
Q
Q
270
|
I
I
Y
Y
P
P
G
G
I
I
K
K
V
V
R
R
Q
Q
L
L
280
|
C
C
K
K
L
L
L
L
R
R
G
G
T
T
K
K
A
A
L
L
290
|
T
T
E
E
V
V
I
M
P
P
L
L
T
T
E
E
E
E
A
A
300
|
E
E
L
L
E
E
L
L
A
A
E
E
N
N
R
R
E
E
I
I
310
|
L
L
K
K
E
E
P
P
V
V
H
H
G
G
V
V
Y
Y
Y
Y
320
|
D
D
P
P
S
S
K
K
D
D
L
L
I
I
A
A
E
E
I
I
330
|
Q
Q
K
K
Q
Q
G
G
Q
Q
G
G
Q
Q
W
W
T
T
Y
Y
340
|
Q
Q
I
I
Y
Y
Q
Q
E
E
P
P
F
F
K
K
N
N
L
L
350
|
K
K
T
T
G
G
K
K
Y
Y
A
A
R
R
M
M
R
R
G
G
360
|
A
A
H
H
T
T
N
N
D
D
V
V
K
K
Q
Q
L
L
T
T
370
|
E
E
A
A
V
V
Q
Q
K
K
I
I
T
T
T
T
E
E
S
S
380
|
I
I
V
V
I
I
W
W
G
G
K
K
T
T
P
P
K
K
F
F
390
|
K
K
L
L
P
P
I
I
Q
Q
K
K
E
E
T
T
W
W
E
E
400
|
T
T
W
W
W
W
T
T
E
E
Y
Y
W
W
Q
Q
A
A
T
T
410
|
W
W
I
I
P
P
E
E
W
W
E
E
F
F
V
V
N
N
T
T
420
|
P
P
P
P
L
L
V
V
K
K
L
L
W
W
Y
Y
Q
Q
L
L
430
|
E
E
K
K
E
E
P
P
I
I
V
V
G
G
A
A
E
E
T
T
440
|
F
F
Y
Y
V
V
D
D
G
G
A
A
A
A
N
N
R
R
E
E
450
|
T
T
K
K
L
L
G
G
K
K
A
A
G
G
Y
Y
V
V
T
T
460
|
N
N
R
R
G
G
R
R
Q
Q
K
K
V
V
V
V
T
T
L
L
470
|
T
T
D
D
T
T
T
T
N
N
Q
Q
K
K
T
T
E
E
L
L
480
|
Q
Q
A
A
I
I
Y
Y
L
L
A
A
L
L
Q
Q
D
D
S
S
490
|
G
G
L
L
E
E
V
V
N
N
I
I
V
V
T
T
D
D
S
S
500
|
Q
Q
Y
Y
A
A
L
L
G
G
I
I
I
I
Q
Q
A
A
Q
Q
510
|
P
P
D
D
Q
Q
S
S
E
E
S
S
E
E
L
L
V
V
N
N
520
|
Q
Q
I
I
I
I
E
E
Q
Q
L
L
I
I
K
K
K
K
E
E
530
|
K
K
V
V
Y
Y
L
L
A
A
W
W
V
V
P
P
A
A
H
H
540
|
K
K
G
G
I
I
G
G
G
G
N
N
E
E
Q
Q
V
V
D
D
550
|
K
K
L
L
V
V
S
S
A
A
G
G
I
I
R
R
K
K
V
V
560
|
L
L
|
|||||||||||||
| Experimental Note | Identified from the Human Clinical Data | ||||||||||||
| Experiment for Molecule Alteration |
Site-directed mutagenesis; Sanger sequencing assay | ||||||||||||
| Experiment for Drug Resistance |
PhenoSense assay | ||||||||||||
| Mechanism Description | Sixteen mutations at eight positions had a mean regression coefficient >=1.0 log10 (a contribution to decreased susceptibility of 10-fold or greater) for nevirapine, efavirenz, etravirine and/or rilpivirine: L100I, k101P, k103N/S, V106A/M, Y181C/I/V, Y188C/L, G190A/E/Q/S and F227C. With the exception of L100I and F227C, each mutation had a mean regression coefficient >=1.0 log10 for nevirapine. With the exception of k103S, V106A, Y181C/I/V and F227C, each had a mean regression coefficient >=1.0 log10 for efavirenz. Five mutations, k101P, Y181I/V, G190E and F227C, had a mean regression coefficient >=1.0 log10 for etravirine. Six mutations, L100I, k101P, Y181I/V, G190E and F227C, had a mean regression coefficient >= 1.0 log10 for rilpivirine. | ||||||||||||
| Key Molecule: HIV1 Reverse transcriptase (HIV1 RT) | [2] | ||||||||||||
| Resistant Disease | Human immunodeficiency virus infection [ICD-11: 1C62.0] | ||||||||||||
| Molecule Alteration | Missense mutation | p.M16MV+p.T69TN+p.V90I+p.L100I+p.K103N+p.D123E+p.I135V+p.G196E+p.P243S+p.V245E+p.E248DV+p.I293V+p.E297R |
|||||||||||
| Wild Type Structure | Method: X-ray diffraction | Resolution: 2.09 Å | |||||||||||
| Mutant Type Structure | Method: AlphaFold | Average pLDDT: 86.682 |

|
||||||||||
| Download The Information of Sequence | Download The Structure File | ||||||||||||
-
P
P
I
I
S
S
P
P
I
I
E
E
T
T
V
V
P
P
10
|
V
V
K
K
L
L
K
K
P
P
G
G
M
V
D
D
G
G
P
P
20
|
K
K
V
V
K
K
Q
Q
W
W
P
P
L
L
T
T
E
E
E
E
30
|
K
K
I
I
K
K
A
A
L
L
V
V
E
E
I
I
C
C
T
T
40
|
E
E
M
M
E
E
K
K
E
E
G
G
K
K
I
I
S
S
K
K
50
|
I
I
G
G
P
P
E
E
N
N
P
P
Y
Y
N
N
T
T
P
P
60
|
V
V
F
F
A
A
I
I
K
K
K
K
K
K
D
D
S
S
T
N
70
|
K
K
W
W
R
R
K
K
L
L
V
V
D
D
F
F
R
R
E
E
80
|
L
L
N
N
K
K
R
R
T
T
Q
Q
D
D
F
F
W
W
E
E
90
|
V
I
Q
Q
L
L
G
G
I
I
P
P
H
H
P
P
A
A
G
G
100
|
L
I
K
K
K
K
K
N
K
K
S
S
V
V
T
T
V
V
L
L
110
|
D
D
V
V
G
G
D
D
A
A
Y
Y
F
F
S
S
V
V
P
P
120
|
L
L
D
D
E
E
D
E
F
F
R
R
K
K
Y
Y
T
T
A
A
130
|
F
F
T
T
I
I
P
P
S
S
I
V
N
N
N
N
E
E
T
T
140
|
P
P
G
G
I
I
R
R
Y
Y
Q
Q
Y
Y
N
N
V
V
L
L
150
|
P
P
Q
Q
G
G
W
W
K
K
G
G
S
S
P
P
A
A
I
I
160
|
F
F
Q
Q
S
S
S
S
M
M
T
T
K
K
I
I
L
L
E
E
170
|
P
P
F
F
R
R
K
K
Q
Q
N
N
P
P
D
D
I
I
V
V
180
|
I
I
Y
Y
Q
Q
Y
Y
M
M
D
D
D
D
L
L
Y
Y
V
V
190
|
G
G
S
S
D
D
L
L
E
E
I
I
G
E
Q
Q
H
H
R
R
200
|
T
T
K
K
I
I
E
E
E
E
L
L
R
R
Q
Q
H
H
L
L
210
|
L
L
R
R
W
W
G
G
L
L
T
T
T
T
P
P
D
D
K
K
220
|
K
K
H
H
Q
Q
K
K
E
E
P
P
P
P
F
F
L
L
W
W
230
|
M
M
G
G
Y
Y
E
E
L
L
H
H
P
P
D
D
K
K
W
W
240
|
T
T
V
V
Q
Q
P
S
I
I
V
E
L
L
P
P
E
D
K
K
250
|
D
D
S
S
W
W
T
T
V
V
N
N
D
D
I
I
Q
Q
K
K
260
|
L
L
V
V
G
G
K
K
L
L
N
N
W
W
A
A
S
S
Q
Q
270
|
I
I
Y
Y
P
P
G
G
I
I
K
K
V
V
R
R
Q
Q
L
L
280
|
C
C
K
K
L
L
L
L
R
R
G
G
T
T
K
K
A
A
L
L
290
|
T
T
E
E
V
V
I
V
P
P
L
L
T
T
E
R
E
E
A
A
300
|
E
E
L
L
E
E
L
L
A
A
E
E
N
N
R
R
E
E
I
I
310
|
L
L
K
K
E
E
P
P
V
V
H
H
G
G
V
V
Y
Y
Y
Y
320
|
D
D
P
P
S
S
K
K
D
D
L
L
I
I
A
A
E
E
I
I
330
|
Q
Q
K
K
Q
Q
G
G
Q
Q
G
G
Q
Q
W
W
T
T
Y
Y
340
|
Q
Q
I
I
Y
Y
Q
Q
E
E
P
P
F
F
K
K
N
N
L
L
350
|
K
K
T
T
G
G
K
K
Y
Y
A
A
R
R
M
M
R
R
G
G
360
|
A
A
H
H
T
T
N
N
D
D
V
V
K
K
Q
Q
L
L
T
T
370
|
E
E
A
A
V
V
Q
Q
K
K
I
I
T
T
T
T
E
E
S
S
380
|
I
I
V
V
I
I
W
W
G
G
K
K
T
T
P
P
K
K
F
F
390
|
K
K
L
L
P
P
I
I
Q
Q
K
K
E
E
T
T
W
W
E
E
400
|
T
T
W
W
W
W
T
T
E
E
Y
Y
W
W
Q
Q
A
A
T
T
410
|
W
W
I
I
P
P
E
E
W
W
E
E
F
F
V
V
N
N
T
T
420
|
P
P
P
P
L
L
V
V
K
K
L
L
W
W
Y
Y
Q
Q
L
L
430
|
E
E
K
K
E
E
P
P
I
I
V
V
G
G
A
A
E
E
T
T
440
|
F
F
Y
Y
V
V
D
D
G
G
A
A
A
A
N
N
R
R
E
E
450
|
T
T
K
K
L
L
G
G
K
K
A
A
G
G
Y
Y
V
V
T
T
460
|
N
N
R
R
G
G
R
R
Q
Q
K
K
V
V
V
V
T
T
L
L
470
|
T
T
D
D
T
T
T
T
N
N
Q
Q
K
K
T
T
E
E
L
L
480
|
Q
Q
A
A
I
I
Y
Y
L
L
A
A
L
L
Q
Q
D
D
S
S
490
|
G
G
L
L
E
E
V
V
N
N
I
I
V
V
T
T
D
D
S
S
500
|
Q
Q
Y
Y
A
A
L
L
G
G
I
I
I
I
Q
Q
A
A
Q
Q
510
|
P
P
D
D
Q
Q
S
S
E
E
S
S
E
E
L
L
V
V
N
N
520
|
Q
Q
I
I
I
I
E
E
Q
Q
L
L
I
I
K
K
K
K
E
E
530
|
K
K
V
V
Y
Y
L
L
A
A
W
W
V
V
P
P
A
A
H
H
540
|
K
K
G
G
I
I
G
G
G
G
N
N
E
E
Q
Q
V
V
D
D
550
|
K
K
L
L
V
V
S
S
A
A
G
G
I
I
R
R
K
K
V
V
560
|
L
L
|
|||||||||||||
| Experimental Note | Identified from the Human Clinical Data | ||||||||||||
| Experiment for Molecule Alteration |
Site-directed mutagenesis; Sanger sequencing assay | ||||||||||||
| Experiment for Drug Resistance |
PhenoSense assay | ||||||||||||
| Mechanism Description | Sixteen mutations at eight positions had a mean regression coefficient >=1.0 log10 (a contribution to decreased susceptibility of 10-fold or greater) for nevirapine, efavirenz, etravirine and/or rilpivirine: L100I, k101P, k103N/S, V106A/M, Y181C/I/V, Y188C/L, G190A/E/Q/S and F227C. With the exception of L100I and F227C, each mutation had a mean regression coefficient >=1.0 log10 for nevirapine. With the exception of k103S, V106A, Y181C/I/V and F227C, each had a mean regression coefficient >=1.0 log10 for efavirenz. Five mutations, k101P, Y181I/V, G190E and F227C, had a mean regression coefficient >=1.0 log10 for etravirine. Six mutations, L100I, k101P, Y181I/V, G190E and F227C, had a mean regression coefficient >= 1.0 log10 for rilpivirine. | ||||||||||||
| Key Molecule: HIV1 Reverse transcriptase (HIV1 RT) | [2] | ||||||||||||
| Resistant Disease | Human immunodeficiency virus infection [ICD-11: 1C62.0] | ||||||||||||
| Molecule Alteration | Missense mutation | p.V60I+p.K64Y+p.D67N+p.K70R+p.V90VI+p.K101P+p.T139M+p.M184V+p.G190A+p.K219Q+p.E224D+p.L228H+p.V245E |
|||||||||||
| Wild Type Structure | Method: X-ray diffraction | Resolution: 2.09 Å | |||||||||||
| Mutant Type Structure | Method: AlphaFold | Average pLDDT: 86.895 |

|
||||||||||
| Download The Information of Sequence | Download The Structure File | ||||||||||||
-
P
P
I
I
590
|
S
S
P
P
I
I
E
E
T
T
V
V
P
P
V
V
K
K
L
L
600
|
K
K
P
P
G
G
M
M
D
D
G
G
P
P
K
K
V
V
K
K
610
|
Q
Q
W
W
P
P
L
L
T
T
E
E
E
E
K
K
I
I
K
K
620
|
A
A
L
L
V
V
E
E
I
I
C
C
T
T
E
E
M
M
E
E
630
|
K
K
E
E
G
G
K
K
I
I
S
S
K
K
I
I
G
G
P
P
640
|
E
E
N
N
P
P
Y
Y
N
N
T
T
P
P
V
I
F
F
A
A
650
|
I
I
K
Y
K
K
K
K
D
N
S
S
T
T
K
R
W
W
R
R
660
|
K
K
L
L
V
V
D
D
F
F
R
R
E
E
L
L
N
N
K
K
670
|
R
R
T
T
Q
Q
D
D
F
F
W
W
E
E
V
I
Q
Q
L
L
680
|
G
G
I
I
P
P
H
H
P
P
A
A
G
G
L
L
K
P
K
K
690
|
K
K
K
K
S
S
V
V
T
T
V
V
L
L
D
D
V
V
G
G
700
|
D
D
A
A
Y
Y
F
F
S
S
V
V
P
P
L
L
D
D
E
E
710
|
D
D
F
F
R
R
K
K
Y
Y
T
T
A
A
F
F
T
T
I
I
720
|
P
P
S
S
I
I
N
N
N
N
E
E
T
M
P
P
G
G
I
I
730
|
R
R
Y
Y
Q
Q
Y
Y
N
N
V
V
L
L
P
P
Q
Q
G
G
740
|
W
W
K
K
G
G
S
S
P
P
A
A
I
I
F
F
Q
Q
S
S
750
|
S
S
M
M
T
T
K
K
I
I
L
L
E
E
P
P
F
F
R
R
760
|
K
K
Q
Q
N
N
P
P
D
D
I
I
V
V
I
I
Y
Y
Q
Q
770
|
Y
Y
M
V
D
D
D
D
L
L
Y
Y
V
V
G
A
S
S
D
D
780
|
L
L
E
E
I
I
G
G
Q
Q
H
H
R
R
T
T
K
K
I
I
790
|
E
E
E
E
L
L
R
R
Q
Q
H
H
L
L
L
L
R
R
W
W
800
|
G
G
L
L
T
T
T
T
P
P
D
D
K
Q
K
K
H
H
Q
Q
810
|
K
K
E
D
P
P
P
P
F
F
L
H
W
W
M
M
G
G
Y
Y
820
|
E
E
L
L
H
H
P
P
D
D
K
K
W
W
T
T
V
V
Q
Q
830
|
P
P
I
I
V
E
L
L
P
P
E
E
K
K
D
D
S
S
W
W
840
|
T
T
V
V
N
N
D
D
I
I
Q
Q
K
K
L
L
V
V
G
G
850
|
K
K
L
L
N
N
W
W
A
A
S
S
Q
Q
I
I
Y
Y
P
P
860
|
G
G
I
I
K
K
V
V
R
R
Q
Q
L
L
C
C
K
K
L
L
870
|
L
L
R
R
G
G
T
T
K
K
A
A
L
L
T
T
E
E
V
V
880
|
I
I
P
P
L
L
T
T
E
E
E
E
A
A
E
E
L
L
E
E
890
|
L
L
A
A
E
E
N
N
R
R
E
E
I
I
L
L
K
K
E
E
900
|
P
P
V
V
H
H
G
G
V
V
Y
Y
Y
Y
D
D
P
P
S
S
910
|
K
K
D
D
L
L
I
I
A
A
E
E
I
I
Q
Q
K
K
Q
Q
920
|
G
G
Q
Q
G
G
Q
Q
W
W
T
T
Y
Y
Q
Q
I
I
Y
Y
930
|
Q
Q
E
E
P
P
F
F
K
K
N
N
L
L
K
K
T
T
G
G
940
|
K
K
Y
Y
A
A
R
R
M
M
R
R
G
G
A
A
H
H
T
T
950
|
N
N
D
D
V
V
K
K
Q
Q
L
L
T
T
E
E
A
A
V
V
960
|
Q
Q
K
K
I
I
T
T
T
T
E
E
S
S
I
I
V
V
I
I
970
|
W
W
G
G
K
K
T
T
P
P
K
K
F
F
K
K
L
L
P
P
980
|
I
I
Q
Q
K
K
E
E
T
T
W
W
E
E
T
T
W
W
W
W
990
|
T
T
E
E
Y
Y
W
W
Q
Q
A
A
T
T
W
W
I
I
P
P
1000
|
E
E
W
W
E
E
F
F
V
V
N
N
T
T
P
P
P
P
L
L
1010
|
V
V
K
K
L
L
W
W
Y
Y
Q
Q
L
L
E
E
K
K
E
E
1020
|
P
P
I
I
V
V
G
G
A
A
E
E
T
T
F
F
Y
Y
V
V
1030
|
D
D
G
G
A
A
A
A
N
N
R
R
E
E
T
T
K
K
L
L
1040
|
G
G
K
K
A
A
G
G
Y
Y
V
V
T
T
N
N
R
R
G
G
1050
|
R
R
Q
Q
K
K
V
V
V
V
T
T
L
L
T
T
D
D
T
T
1060
|
T
T
N
N
Q
Q
K
K
T
T
E
E
L
L
Q
Q
A
A
I
I
1070
|
Y
Y
L
L
A
A
L
L
Q
Q
D
D
S
S
G
G
L
L
E
E
1080
|
V
V
N
N
I
I
V
V
T
T
D
D
S
S
Q
Q
Y
Y
A
A
1090
|
L
L
G
G
I
I
I
I
Q
Q
A
A
Q
Q
P
P
D
D
Q
Q
1100
|
S
S
E
E
S
S
E
E
L
L
V
V
N
N
Q
Q
I
I
I
I
1110
|
E
E
Q
Q
L
L
I
I
K
K
K
K
E
E
K
K
V
V
Y
Y
1120
|
L
L
A
A
W
W
V
V
P
P
A
A
H
H
K
K
G
G
I
I
1130
|
G
G
G
G
N
N
E
E
Q
Q
V
V
D
D
K
K
L
L
V
V
1140
|
S
S
A
A
G
G
I
I
R
R
K
K
V
V
L
L
|
|||||||||||||
| Experimental Note | Identified from the Human Clinical Data | ||||||||||||
| Experiment for Molecule Alteration |
Site-directed mutagenesis; Sanger sequencing assay | ||||||||||||
| Experiment for Drug Resistance |
PhenoSense assay | ||||||||||||
| Mechanism Description | Sixteen mutations at eight positions had a mean regression coefficient >=1.0 log10 (a contribution to decreased susceptibility of 10-fold or greater) for nevirapine, efavirenz, etravirine and/or rilpivirine: L100I, k101P, k103N/S, V106A/M, Y181C/I/V, Y188C/L, G190A/E/Q/S and F227C. With the exception of L100I and F227C, each mutation had a mean regression coefficient >=1.0 log10 for nevirapine. With the exception of k103S, V106A, Y181C/I/V and F227C, each had a mean regression coefficient >=1.0 log10 for efavirenz. Five mutations, k101P, Y181I/V, G190E and F227C, had a mean regression coefficient >=1.0 log10 for etravirine. Six mutations, L100I, k101P, Y181I/V, G190E and F227C, had a mean regression coefficient >= 1.0 log10 for rilpivirine. | ||||||||||||
| Key Molecule: HIV1 Reverse transcriptase (HIV1 RT) | [2] | ||||||||||||
| Resistant Disease | Human immunodeficiency virus infection [ICD-11: 1C62.0] | ||||||||||||
| Molecule Alteration | Missense mutation | p.M41L+p.L74V+p.I142V+p.T165L+p.E169D+p.Y181I+p.M184V+p.R211K+p.T215Y |
|||||||||||
| Wild Type Structure | Method: X-ray diffraction | Resolution: 2.09 Å | |||||||||||
| Mutant Type Structure | Method: AlphaFold | Average pLDDT: 86.827 |

|
||||||||||
| Download The Information of Sequence | Download The Structure File | ||||||||||||
-
P
P
I
I
590
|
S
S
P
P
I
I
E
E
T
T
V
V
P
P
V
V
K
K
L
L
600
|
K
K
P
P
G
G
M
M
D
D
G
G
P
P
K
K
V
V
K
K
610
|
Q
Q
W
W
P
P
L
L
T
T
E
E
E
E
K
K
I
I
K
K
620
|
A
A
L
L
V
V
E
E
I
I
C
C
T
T
E
E
M
L
E
E
630
|
K
K
E
E
G
G
K
K
I
I
S
S
K
K
I
I
G
G
P
P
640
|
E
E
N
N
P
P
Y
Y
N
N
T
T
P
P
V
V
F
F
A
A
650
|
I
I
K
K
K
K
K
K
D
D
S
S
T
T
K
K
W
W
R
R
660
|
K
K
L
V
V
V
D
D
F
F
R
R
E
E
L
L
N
N
K
K
670
|
R
R
T
T
Q
Q
D
D
F
F
W
W
E
E
V
V
Q
Q
L
L
680
|
G
G
I
I
P
P
H
H
P
P
A
A
G
G
L
L
K
K
K
K
690
|
K
K
K
K
S
S
V
V
T
T
V
V
L
L
D
D
V
V
G
G
700
|
D
D
A
A
Y
Y
F
F
S
S
V
V
P
P
L
L
D
D
E
E
710
|
D
D
F
F
R
R
K
K
Y
Y
T
T
A
A
F
F
T
T
I
I
720
|
P
P
S
S
I
I
N
N
N
N
E
E
T
T
P
P
G
G
I
V
730
|
R
R
Y
Y
Q
Q
Y
Y
N
N
V
V
L
L
P
P
Q
Q
G
G
740
|
W
W
K
K
G
G
S
S
P
P
A
A
I
I
F
F
Q
Q
S
S
750
|
S
S
M
M
T
L
K
K
I
I
L
L
E
D
P
P
F
F
R
R
760
|
K
K
Q
Q
N
N
P
P
D
D
I
I
V
V
I
I
Y
I
Q
Q
770
|
Y
Y
M
V
D
D
D
D
L
L
Y
Y
V
V
G
G
S
S
D
D
780
|
L
L
E
E
I
I
G
G
Q
Q
H
H
R
R
T
T
K
K
I
I
790
|
E
E
E
E
L
L
R
R
Q
Q
H
H
L
L
L
L
R
K
W
W
800
|
G
G
L
L
T
Y
T
T
P
P
D
D
K
K
K
K
H
H
Q
Q
810
|
K
K
E
E
P
P
P
P
F
F
L
L
W
W
M
M
G
G
Y
Y
820
|
E
E
L
L
H
H
P
P
D
D
K
K
W
W
T
T
V
V
Q
Q
830
|
P
P
I
I
V
V
L
L
P
P
E
E
K
K
D
D
S
S
W
W
840
|
T
T
V
V
N
N
D
D
I
I
Q
Q
K
K
L
L
V
V
G
G
850
|
K
K
L
L
N
N
W
W
A
A
S
S
Q
Q
I
I
Y
Y
P
P
860
|
G
G
I
I
K
K
V
V
R
R
Q
Q
L
L
C
C
K
K
L
L
870
|
L
L
R
R
G
G
T
T
K
K
A
A
L
L
T
T
E
E
V
V
880
|
I
I
P
P
L
L
T
T
E
E
E
E
A
A
E
E
L
L
E
E
890
|
L
L
A
A
E
E
N
N
R
R
E
E
I
I
L
L
K
K
E
E
900
|
P
P
V
V
H
H
G
G
V
V
Y
Y
Y
Y
D
D
P
P
S
S
910
|
K
K
D
D
L
L
I
I
A
A
E
E
I
I
Q
Q
K
K
Q
Q
920
|
G
G
Q
Q
G
G
Q
Q
W
W
T
T
Y
Y
Q
Q
I
I
Y
Y
930
|
Q
Q
E
E
P
P
F
F
K
K
N
N
L
L
K
K
T
T
G
G
940
|
K
K
Y
Y
A
A
R
R
M
M
R
R
G
G
A
A
H
H
T
T
950
|
N
N
D
D
V
V
K
K
Q
Q
L
L
T
T
E
E
A
A
V
V
960
|
Q
Q
K
K
I
I
T
T
T
T
E
E
S
S
I
I
V
V
I
I
970
|
W
W
G
G
K
K
T
T
P
P
K
K
F
F
K
K
L
L
P
P
980
|
I
I
Q
Q
K
K
E
E
T
T
W
W
E
E
T
T
W
W
W
W
990
|
T
T
E
E
Y
Y
W
W
Q
Q
A
A
T
T
W
W
I
I
P
P
1000
|
E
E
W
W
E
E
F
F
V
V
N
N
T
T
P
P
P
P
L
L
1010
|
V
V
K
K
L
L
W
W
Y
Y
Q
Q
L
L
E
E
K
K
E
E
1020
|
P
P
I
I
V
V
G
G
A
A
E
E
T
T
F
F
Y
Y
V
V
1030
|
D
D
G
G
A
A
A
A
N
N
R
R
E
E
T
T
K
K
L
L
1040
|
G
G
K
K
A
A
G
G
Y
Y
V
V
T
T
N
N
R
R
G
G
1050
|
R
R
Q
Q
K
K
V
V
V
V
T
T
L
L
T
T
D
D
T
T
1060
|
T
T
N
N
Q
Q
K
K
T
T
E
E
L
L
Q
Q
A
A
I
I
1070
|
Y
Y
L
L
A
A
L
L
Q
Q
D
D
S
S
G
G
L
L
E
E
1080
|
V
V
N
N
I
I
V
V
T
T
D
D
S
S
Q
Q
Y
Y
A
A
1090
|
L
L
G
G
I
I
I
I
Q
Q
A
A
Q
Q
P
P
D
D
Q
Q
1100
|
S
S
E
E
S
S
E
E
L
L
V
V
N
N
Q
Q
I
I
I
I
1110
|
E
E
Q
Q
L
L
I
I
K
K
K
K
E
E
K
K
V
V
Y
Y
1120
|
L
L
A
A
W
W
V
V
P
P
A
A
H
H
K
K
G
G
I
I
1130
|
G
G
G
G
N
N
E
E
Q
Q
V
V
D
D
K
K
L
L
V
V
1140
|
S
S
A
A
G
G
I
I
R
R
K
K
V
V
L
L
|
|||||||||||||
| Experimental Note | Identified from the Human Clinical Data | ||||||||||||
| Experiment for Molecule Alteration |
Site-directed mutagenesis; Sanger sequencing assay | ||||||||||||
| Experiment for Drug Resistance |
PhenoSense assay | ||||||||||||
| Mechanism Description | Sixteen mutations at eight positions had a mean regression coefficient >=1.0 log10 (a contribution to decreased susceptibility of 10-fold or greater) for nevirapine, efavirenz, etravirine and/or rilpivirine: L100I, k101P, k103N/S, V106A/M, Y181C/I/V, Y188C/L, G190A/E/Q/S and F227C. With the exception of L100I and F227C, each mutation had a mean regression coefficient >=1.0 log10 for nevirapine. With the exception of k103S, V106A, Y181C/I/V and F227C, each had a mean regression coefficient >=1.0 log10 for efavirenz. Five mutations, k101P, Y181I/V, G190E and F227C, had a mean regression coefficient >=1.0 log10 for etravirine. Six mutations, L100I, k101P, Y181I/V, G190E and F227C, had a mean regression coefficient >= 1.0 log10 for rilpivirine. | ||||||||||||
| Key Molecule: HIV1 Reverse transcriptase (HIV1 RT) | [2] | ||||||||||||
| Resistant Disease | Human immunodeficiency virus infection [ICD-11: 1C62.0] | ||||||||||||
| Molecule Alteration | Missense mutation | p.M41L+p.E44D+p.D67N+p.T69D+p.K70KR+p.A98S+p.Y181I+p.L210W+p.R211K+p.T215Y+p.D218DE |
|||||||||||
| Wild Type Structure | Method: X-ray diffraction | Resolution: 2.09 Å | |||||||||||
| Mutant Type Structure | Method: AlphaFold | Average pLDDT: 87.394 |

|
||||||||||
| Download The Information of Sequence | Download The Structure File | ||||||||||||
-
P
P
I
I
590
|
S
S
P
P
I
I
E
E
T
T
V
V
P
P
V
V
K
K
L
L
600
|
K
K
P
P
G
G
M
M
D
D
G
G
P
P
K
K
V
V
K
K
610
|
Q
Q
W
W
P
P
L
L
T
T
E
E
E
E
K
K
I
I
K
K
620
|
A
A
L
L
V
V
E
E
I
I
C
C
T
T
E
E
M
L
E
E
630
|
K
K
E
D
G
G
K
K
I
I
S
S
K
K
I
I
G
G
P
P
640
|
E
E
N
N
P
P
Y
Y
N
N
T
T
P
P
V
V
F
F
A
A
650
|
I
I
K
K
K
K
K
K
D
N
S
S
T
D
K
R
W
W
R
R
660
|
K
K
L
L
V
V
D
D
F
F
R
R
E
E
L
L
N
N
K
K
670
|
R
R
T
T
Q
Q
D
D
F
F
W
W
E
E
V
V
Q
Q
L
L
680
|
G
G
I
I
P
P
H
H
P
P
A
S
G
G
L
L
K
K
K
K
690
|
K
K
K
K
S
S
V
V
T
T
V
V
L
L
D
D
V
V
G
G
700
|
D
D
A
A
Y
Y
F
F
S
S
V
V
P
P
L
L
D
D
E
E
710
|
D
D
F
F
R
R
K
K
Y
Y
T
T
A
A
F
F
T
T
I
I
720
|
P
P
S
S
I
I
N
N
N
N
E
E
T
T
P
P
G
G
I
I
730
|
R
R
Y
Y
Q
Q
Y
Y
N
N
V
V
L
L
P
P
Q
Q
G
G
740
|
W
W
K
K
G
G
S
S
P
P
A
A
I
I
F
F
Q
Q
S
S
750
|
S
S
M
M
T
T
K
K
I
I
L
L
E
E
P
P
F
F
R
R
760
|
K
K
Q
Q
N
N
P
P
D
D
I
I
V
V
I
I
Y
I
Q
Q
770
|
Y
Y
M
M
D
D
D
D
L
L
Y
Y
V
V
G
G
S
S
D
D
780
|
L
L
E
E
I
I
G
G
Q
Q
H
H
R
R
T
T
K
K
I
I
790
|
E
E
E
E
L
L
R
R
Q
Q
H
H
L
L
L
W
R
K
W
W
800
|
G
G
L
L
T
Y
T
T
P
P
D
E
K
K
K
K
H
H
Q
Q
810
|
K
K
E
E
P
P
P
P
F
F
L
L
W
W
M
M
G
G
Y
Y
820
|
E
E
L
L
H
H
P
P
D
D
K
K
W
W
T
T
V
V
Q
Q
830
|
P
P
I
I
V
V
L
L
P
P
E
E
K
K
D
D
S
S
W
W
840
|
T
T
V
V
N
N
D
D
I
I
Q
Q
K
K
L
L
V
V
G
G
850
|
K
K
L
L
N
N
W
W
A
A
S
S
Q
Q
I
I
Y
Y
P
P
860
|
G
G
I
I
K
K
V
V
R
R
Q
Q
L
L
C
C
K
K
L
L
870
|
L
L
R
R
G
G
T
T
K
K
A
A
L
L
T
T
E
E
V
V
880
|
I
I
P
P
L
L
T
T
E
E
E
E
A
A
E
E
L
L
E
E
890
|
L
L
A
A
E
E
N
N
R
R
E
E
I
I
L
L
K
K
E
E
900
|
P
P
V
V
H
H
G
G
V
V
Y
Y
Y
Y
D
D
P
P
S
S
910
|
K
K
D
D
L
L
I
I
A
A
E
E
I
I
Q
Q
K
K
Q
Q
920
|
G
G
Q
Q
G
G
Q
Q
W
W
T
T
Y
Y
Q
Q
I
I
Y
Y
930
|
Q
Q
E
E
P
P
F
F
K
K
N
N
L
L
K
K
T
T
G
G
940
|
K
K
Y
Y
A
A
R
R
M
M
R
R
G
G
A
A
H
H
T
T
950
|
N
N
D
D
V
V
K
K
Q
Q
L
L
T
T
E
E
A
A
V
V
960
|
Q
Q
K
K
I
I
T
T
T
T
E
E
S
S
I
I
V
V
I
I
970
|
W
W
G
G
K
K
T
T
P
P
K
K
F
F
K
K
L
L
P
P
980
|
I
I
Q
Q
K
K
E
E
T
T
W
W
E
E
T
T
W
W
W
W
990
|
T
T
E
E
Y
Y
W
W
Q
Q
A
A
T
T
W
W
I
I
P
P
1000
|
E
E
W
W
E
E
F
F
V
V
N
N
T
T
P
P
P
P
L
L
1010
|
V
V
K
K
L
L
W
W
Y
Y
Q
Q
L
L
E
E
K
K
E
E
1020
|
P
P
I
I
V
V
G
G
A
A
E
E
T
T
F
F
Y
Y
V
V
1030
|
D
D
G
G
A
A
A
A
N
N
R
R
E
E
T
T
K
K
L
L
1040
|
G
G
K
K
A
A
G
G
Y
Y
V
V
T
T
N
N
R
R
G
G
1050
|
R
R
Q
Q
K
K
V
V
V
V
T
T
L
L
T
T
D
D
T
T
1060
|
T
T
N
N
Q
Q
K
K
T
T
E
E
L
L
Q
Q
A
A
I
I
1070
|
Y
Y
L
L
A
A
L
L
Q
Q
D
D
S
S
G
G
L
L
E
E
1080
|
V
V
N
N
I
I
V
V
T
T
D
D
S
S
Q
Q
Y
Y
A
A
1090
|
L
L
G
G
I
I
I
I
Q
Q
A
A
Q
Q
P
P
D
D
Q
Q
1100
|
S
S
E
E
S
S
E
E
L
L
V
V
N
N
Q
Q
I
I
I
I
1110
|
E
E
Q
Q
L
L
I
I
K
K
K
K
E
E
K
K
V
V
Y
Y
1120
|
L
L
A
A
W
W
V
V
P
P
A
A
H
H
K
K
G
G
I
I
1130
|
G
G
G
G
N
N
E
E
Q
Q
V
V
D
D
K
K
L
L
V
V
1140
|
S
S
A
A
G
G
I
I
R
R
K
K
V
V
L
L
|
|||||||||||||
| Experimental Note | Identified from the Human Clinical Data | ||||||||||||
| Experiment for Molecule Alteration |
Site-directed mutagenesis; Sanger sequencing assay | ||||||||||||
| Experiment for Drug Resistance |
PhenoSense assay | ||||||||||||
| Mechanism Description | Sixteen mutations at eight positions had a mean regression coefficient >=1.0 log10 (a contribution to decreased susceptibility of 10-fold or greater) for nevirapine, efavirenz, etravirine and/or rilpivirine: L100I, k101P, k103N/S, V106A/M, Y181C/I/V, Y188C/L, G190A/E/Q/S and F227C. With the exception of L100I and F227C, each mutation had a mean regression coefficient >=1.0 log10 for nevirapine. With the exception of k103S, V106A, Y181C/I/V and F227C, each had a mean regression coefficient >=1.0 log10 for efavirenz. Five mutations, k101P, Y181I/V, G190E and F227C, had a mean regression coefficient >=1.0 log10 for etravirine. Six mutations, L100I, k101P, Y181I/V, G190E and F227C, had a mean regression coefficient >= 1.0 log10 for rilpivirine. | ||||||||||||
| Key Molecule: HIV1 Reverse transcriptase (HIV1 RT) | [4] | ||||||||||||
| Resistant Disease | Human immunodeficiency virus infection [ICD-11: 1C62.0] | ||||||||||||
| Molecule Alteration | Missense mutation | p.K20R+p.K32KQ+p.M41L+p.E53ED+p.P59PS+p.L74V+p.I135T+p.Q145QR+p.Y181V+p.R211K+p.T215Y+p.K249R |
|||||||||||
| Wild Type Structure | Method: X-ray diffraction | Resolution: 2.09 Å | |||||||||||
| Mutant Type Structure | Method: AlphaFold | Average pLDDT: 86.709 |

|
||||||||||
| Download The Information of Sequence | Download The Structure File | ||||||||||||
-
P
P
I
I
590
|
S
S
P
P
I
I
E
E
T
T
V
V
P
P
V
V
K
K
L
L
600
|
K
K
P
P
G
G
M
M
D
D
G
G
P
P
K
R
V
V
K
K
610
|
Q
Q
W
W
P
P
L
L
T
T
E
E
E
E
K
K
I
I
K
Q
620
|
A
A
L
L
V
V
E
E
I
I
C
C
T
T
E
E
M
L
E
E
630
|
K
K
E
E
G
G
K
K
I
I
S
S
K
K
I
I
G
G
P
P
640
|
E
D
N
N
P
P
Y
Y
N
N
T
T
P
S
V
V
F
F
A
A
650
|
I
I
K
K
K
K
K
K
D
D
S
S
T
T
K
K
W
W
R
R
660
|
K
K
L
V
V
V
D
D
F
F
R
R
E
E
L
L
N
N
K
K
670
|
R
R
T
T
Q
Q
D
D
F
F
W
W
E
E
V
V
Q
Q
L
L
680
|
G
G
I
I
P
P
H
H
P
P
A
A
G
G
L
L
K
K
K
K
690
|
K
K
K
K
S
S
V
V
T
T
V
V
L
L
D
D
V
V
G
G
700
|
D
D
A
A
Y
Y
F
F
S
S
V
V
P
P
L
L
D
D
E
E
710
|
D
D
F
F
R
R
K
K
Y
Y
T
T
A
A
F
F
T
T
I
I
720
|
P
P
S
S
I
T
N
N
N
N
E
E
T
T
P
P
G
G
I
I
730
|
R
R
Y
Y
Q
R
Y
Y
N
N
V
V
L
L
P
P
Q
Q
G
G
740
|
W
W
K
K
G
G
S
S
P
P
A
A
I
I
F
F
Q
Q
S
S
750
|
S
S
M
M
T
T
K
K
I
I
L
L
E
E
P
P
F
F
R
R
760
|
K
K
Q
Q
N
N
P
P
D
D
I
I
V
V
I
I
Y
V
Q
Q
770
|
Y
Y
M
M
D
D
D
D
L
L
Y
Y
V
V
G
G
S
S
D
D
780
|
L
L
E
E
I
I
G
G
Q
Q
H
H
R
R
T
T
K
K
I
I
790
|
E
E
E
E
L
L
R
R
Q
Q
H
H
L
L
L
L
R
K
W
W
800
|
G
G
L
L
T
Y
T
T
P
P
D
D
K
K
K
K
H
H
Q
Q
810
|
K
K
E
E
P
P
P
P
F
F
L
L
W
W
M
M
G
G
Y
Y
820
|
E
E
L
L
H
H
P
P
D
D
K
K
W
W
T
T
V
V
Q
Q
830
|
P
P
I
I
V
V
L
L
P
P
E
E
K
R
D
D
S
S
W
W
840
|
T
T
V
V
N
N
D
D
I
I
Q
Q
K
K
L
L
V
V
G
G
850
|
K
K
L
L
N
N
W
W
A
A
S
S
Q
Q
I
I
Y
Y
P
P
860
|
G
G
I
I
K
K
V
V
R
R
Q
Q
L
L
C
C
K
K
L
L
870
|
L
L
R
R
G
G
T
T
K
K
A
A
L
L
T
T
E
E
V
V
880
|
I
I
P
P
L
L
T
T
E
E
E
E
A
A
E
E
L
L
E
E
890
|
L
L
A
A
E
E
N
N
R
R
E
E
I
I
L
L
K
K
E
E
900
|
P
P
V
V
H
H
G
G
V
V
Y
Y
Y
Y
D
D
P
P
S
S
910
|
K
K
D
D
L
L
I
I
A
A
E
E
I
I
Q
Q
K
K
Q
Q
920
|
G
G
Q
Q
G
G
Q
Q
W
W
T
T
Y
Y
Q
Q
I
I
Y
Y
930
|
Q
Q
E
E
P
P
F
F
K
K
N
N
L
L
K
K
T
T
G
G
940
|
K
K
Y
Y
A
A
R
R
M
M
R
R
G
G
A
A
H
H
T
T
950
|
N
N
D
D
V
V
K
K
Q
Q
L
L
T
T
E
E
A
A
V
V
960
|
Q
Q
K
K
I
I
T
T
T
T
E
E
S
S
I
I
V
V
I
I
970
|
W
W
G
G
K
K
T
T
P
P
K
K
F
F
K
K
L
L
P
P
980
|
I
I
Q
Q
K
K
E
E
T
T
W
W
E
E
T
T
W
W
W
W
990
|
T
T
E
E
Y
Y
W
W
Q
Q
A
A
T
T
W
W
I
I
P
P
1000
|
E
E
W
W
E
E
F
F
V
V
N
N
T
T
P
P
P
P
L
L
1010
|
V
V
K
K
L
L
W
W
Y
Y
Q
Q
L
L
E
E
K
K
E
E
1020
|
P
P
I
I
V
V
G
G
A
A
E
E
T
T
F
F
Y
Y
V
V
1030
|
D
D
G
G
A
A
A
A
N
N
R
R
E
E
T
T
K
K
L
L
1040
|
G
G
K
K
A
A
G
G
Y
Y
V
V
T
T
N
N
R
R
G
G
1050
|
R
R
Q
Q
K
K
V
V
V
V
T
T
L
L
T
T
D
D
T
T
1060
|
T
T
N
N
Q
Q
K
K
T
T
E
E
L
L
Q
Q
A
A
I
I
1070
|
Y
Y
L
L
A
A
L
L
Q
Q
D
D
S
S
G
G
L
L
E
E
1080
|
V
V
N
N
I
I
V
V
T
T
D
D
S
S
Q
Q
Y
Y
A
A
1090
|
L
L
G
G
I
I
I
I
Q
Q
A
A
Q
Q
P
P
D
D
Q
Q
1100
|
S
S
E
E
S
S
E
E
L
L
V
V
N
N
Q
Q
I
I
I
I
1110
|
E
E
Q
Q
L
L
I
I
K
K
K
K
E
E
K
K
V
V
Y
Y
1120
|
L
L
A
A
W
W
V
V
P
P
A
A
H
H
K
K
G
G
I
I
1130
|
G
G
G
G
N
N
E
E
Q
Q
V
V
D
D
K
K
L
L
V
V
1140
|
S
S
A
A
G
G
I
I
R
R
K
K
V
V
L
L
|
|||||||||||||
| Experimental Note | Identified from the Human Clinical Data | ||||||||||||
| Experiment for Molecule Alteration |
Sanger sequencing assay; GeneSeq assay; Population sequencing of the integrase region assay; DNA sequencing assay; Site-directed mutagenesis | ||||||||||||
| Experiment for Drug Resistance |
PhenoSense assay; Viral load assay; A single-cycle assay; TZM-bl cell line-based phenotypic assay; SDMs and phenotyping assay | ||||||||||||
| Mechanism Description | Some mutations identified were assiacted with resistance in the HIV isolates, those mutations included K20R, K32KQ, M41L, E53ED, P59PS, L74V, I135T, Q145QR, Y181V, R211K, T215Y, K249R. | ||||||||||||
| Key Molecule: HIV1 Reverse transcriptase (HIV1 RT) | [5] | ||||||||||||
| Resistant Disease | Human immunodeficiency virus infection [ICD-11: 1C62.0] | ||||||||||||
| Molecule Alteration | Missense mutation | p.V60VI+p.D67G+p.S68G+p.T69N+p.K70R+p.K101P+p.K102Q+p.K103S+p.I142IV+p.D177E+p.I178L+p.M184V+p.T200A+p.T215V+p.K219E+p.I293V+p.E297K |
|||||||||||
| Wild Type Structure | Method: X-ray diffraction | Resolution: 2.09 Å | |||||||||||
| Mutant Type Structure | Method: AlphaFold | Average pLDDT: 87.065 |

|
||||||||||
| Download The Information of Sequence | Download The Structure File | ||||||||||||
-
P
P
I
I
590
|
S
S
P
P
I
I
E
E
T
T
V
V
P
P
V
V
K
K
L
L
600
|
K
K
P
P
G
G
M
M
D
D
G
G
P
P
K
K
V
V
K
K
610
|
Q
Q
W
W
P
P
L
L
T
T
E
E
E
E
K
K
I
I
K
K
620
|
A
A
L
L
V
V
E
E
I
I
C
C
T
T
E
E
M
M
E
E
630
|
K
K
E
E
G
G
K
K
I
I
S
S
K
K
I
I
G
G
P
P
640
|
E
E
N
N
P
P
Y
Y
N
N
T
T
P
P
V
I
F
F
A
A
650
|
I
I
K
K
K
K
K
K
D
G
S
G
T
N
K
R
W
W
R
R
660
|
K
K
L
L
V
V
D
D
F
F
R
R
E
E
L
L
N
N
K
K
670
|
R
R
T
T
Q
Q
D
D
F
F
W
W
E
E
V
V
Q
Q
L
L
680
|
G
G
I
I
P
P
H
H
P
P
A
A
G
G
L
L
K
P
K
Q
690
|
K
S
K
K
S
S
V
V
T
T
V
V
L
L
D
D
V
V
G
G
700
|
D
D
A
A
Y
Y
F
F
S
S
V
V
P
P
L
L
D
D
E
E
710
|
D
D
F
F
R
R
K
K
Y
Y
T
T
A
A
F
F
T
T
I
I
720
|
P
P
S
S
I
I
N
N
N
N
E
E
T
T
P
P
G
G
I
V
730
|
R
R
Y
Y
Q
Q
Y
Y
N
N
V
V
L
L
P
P
Q
Q
G
G
740
|
W
W
K
K
G
G
S
S
P
P
A
A
I
I
F
F
Q
Q
S
S
750
|
S
S
M
M
T
T
K
K
I
I
L
L
E
E
P
P
F
F
R
R
760
|
K
K
Q
Q
N
N
P
P
D
E
I
L
V
V
I
I
Y
Y
Q
Q
770
|
Y
Y
M
V
D
D
D
D
L
L
Y
Y
V
V
G
G
S
S
D
D
780
|
L
L
E
E
I
I
G
G
Q
Q
H
H
R
R
T
A
K
K
I
I
790
|
E
E
E
E
L
L
R
R
Q
Q
H
H
L
L
L
L
R
R
W
W
800
|
G
G
L
L
T
V
T
T
P
P
D
D
K
E
K
K
H
H
Q
Q
810
|
K
K
E
E
P
P
P
P
F
F
L
L
W
W
M
M
G
G
Y
Y
820
|
E
E
L
L
H
H
P
P
D
D
K
K
W
W
T
T
V
V
Q
Q
830
|
P
P
I
I
V
V
L
L
P
P
E
E
K
K
D
D
S
S
W
W
840
|
T
T
V
V
N
N
D
D
I
I
Q
Q
K
K
L
L
V
V
G
G
850
|
K
K
L
L
N
N
W
W
A
A
S
S
Q
Q
I
I
Y
Y
P
P
860
|
G
G
I
I
K
K
V
V
R
R
Q
Q
L
L
C
C
K
K
L
L
870
|
L
L
R
R
G
G
T
T
K
K
A
A
L
L
T
T
E
E
V
V
880
|
I
V
P
P
L
L
T
T
E
K
E
E
A
A
E
E
L
L
E
E
890
|
L
L
A
A
E
E
N
N
R
R
E
E
I
I
L
L
K
K
E
E
900
|
P
P
V
V
H
H
G
G
V
V
Y
Y
Y
Y
D
D
P
P
S
S
910
|
K
K
D
D
L
L
I
I
A
A
E
E
I
I
Q
Q
K
K
Q
Q
920
|
G
G
Q
Q
G
G
Q
Q
W
W
T
T
Y
Y
Q
Q
I
I
Y
Y
930
|
Q
Q
E
E
P
P
F
F
K
K
N
N
L
L
K
K
T
T
G
G
940
|
K
K
Y
Y
A
A
R
R
M
M
R
R
G
G
A
A
H
H
T
T
950
|
N
N
D
D
V
V
K
K
Q
Q
L
L
T
T
E
E
A
A
V
V
960
|
Q
Q
K
K
I
I
T
T
T
T
E
E
S
S
I
I
V
V
I
I
970
|
W
W
G
G
K
K
T
T
P
P
K
K
F
F
K
K
L
L
P
P
980
|
I
I
Q
Q
K
K
E
E
T
T
W
W
E
E
T
T
W
W
W
W
990
|
T
T
E
E
Y
Y
W
W
Q
Q
A
A
T
T
W
W
I
I
P
P
1000
|
E
E
W
W
E
E
F
F
V
V
N
N
T
T
P
P
P
P
L
L
1010
|
V
V
K
K
L
L
W
W
Y
Y
Q
Q
L
L
E
E
K
K
E
E
1020
|
P
P
I
I
V
V
G
G
A
A
E
E
T
T
F
F
Y
Y
V
V
1030
|
D
D
G
G
A
A
A
A
N
N
R
R
E
E
T
T
K
K
L
L
1040
|
G
G
K
K
A
A
G
G
Y
Y
V
V
T
T
N
N
R
R
G
G
1050
|
R
R
Q
Q
K
K
V
V
V
V
T
T
L
L
T
T
D
D
T
T
1060
|
T
T
N
N
Q
Q
K
K
T
T
E
E
L
L
Q
Q
A
A
I
I
1070
|
Y
Y
L
L
A
A
L
L
Q
Q
D
D
S
S
G
G
L
L
E
E
1080
|
V
V
N
N
I
I
V
V
T
T
D
D
S
S
Q
Q
Y
Y
A
A
1090
|
L
L
G
G
I
I
I
I
Q
Q
A
A
Q
Q
P
P
D
D
Q
Q
1100
|
S
S
E
E
S
S
E
E
L
L
V
V
N
N
Q
Q
I
I
I
I
1110
|
E
E
Q
Q
L
L
I
I
K
K
K
K
E
E
K
K
V
V
Y
Y
1120
|
L
L
A
A
W
W
V
V
P
P
A
A
H
H
K
K
G
G
I
I
1130
|
G
G
G
G
N
N
E
E
Q
Q
V
V
D
D
K
K
L
L
V
V
1140
|
S
S
A
A
G
G
I
I
R
R
K
K
V
V
L
L
|
|||||||||||||
| Experimental Note | Identified from the Human Clinical Data | ||||||||||||
| Experiment for Molecule Alteration |
Sanger sequencing assay; GeneSeq assay; Population sequencing of the integrase region assay; DNA sequencing assay; Site-directed mutagenesis | ||||||||||||
| Experiment for Drug Resistance |
PhenoSense assay; Viral load assay; A single-cycle assay; TZM-bl cell line-based phenotypic assay; SDMs and phenotyping assay | ||||||||||||
| Mechanism Description | Some mutations identified were assiacted with resistance in the HIV isolates, those mutations included V60VI, D67G, S68G, T69N, K70R, K101P, K102Q, K103S, I142IV, D177E, I178L, M184V, T200A, T215V, K219E, I293V, E297K. | ||||||||||||
| Key Molecule: HIV1 Reverse transcriptase (HIV1 RT) | [6] | ||||||||||||
| Resistant Disease | Human immunodeficiency virus infection [ICD-11: 1C62.0] | ||||||||||||
| Molecule Alteration | Missense mutation | p.D67N+p.T69N+p.K70R+p.V90I+p.K103N+p.V118I+p.I135M |
|||||||||||
| Wild Type Structure | Method: X-ray diffraction | Resolution: 2.09 Å | |||||||||||
| Mutant Type Structure | Method: AlphaFold | Average pLDDT: 87.147 |

|
||||||||||
| Download The Information of Sequence | Download The Structure File | ||||||||||||
-
P
P
I
I
590
|
S
S
P
P
I
I
E
E
T
T
V
V
P
P
V
V
K
K
L
L
600
|
K
K
P
P
G
G
M
M
D
D
G
G
P
P
K
K
V
V
K
K
610
|
Q
Q
W
W
P
P
L
L
T
T
E
E
E
E
K
K
I
I
K
K
620
|
A
A
L
L
V
V
E
E
I
I
C
C
T
T
E
E
M
M
E
E
630
|
K
K
E
E
G
G
K
K
I
I
S
S
K
K
I
I
G
G
P
P
640
|
E
E
N
N
P
P
Y
Y
N
N
T
T
P
P
V
V
F
F
A
A
650
|
I
I
K
K
K
K
K
K
D
N
S
S
T
N
K
R
W
W
R
R
660
|
K
K
L
L
V
V
D
D
F
F
R
R
E
E
L
L
N
N
K
K
670
|
R
R
T
T
Q
Q
D
D
F
F
W
W
E
E
V
I
Q
Q
L
L
680
|
G
G
I
I
P
P
H
H
P
P
A
A
G
G
L
L
K
K
K
K
690
|
K
N
K
K
S
S
V
V
T
T
V
V
L
L
D
D
V
V
G
G
700
|
D
D
A
A
Y
Y
F
F
S
S
V
I
P
P
L
L
D
D
E
E
710
|
D
D
F
F
R
R
K
K
Y
Y
T
T
A
A
F
F
T
T
I
I
720
|
P
P
S
S
I
M
N
N
N
N
E
E
T
T
P
P
G
G
I
I
730
|
R
R
Y
Y
Q
Q
Y
Y
N
N
V
V
L
L
P
P
Q
Q
G
G
740
|
W
W
K
K
G
G
S
S
P
P
A
A
I
I
F
F
Q
Q
S
S
750
|
S
S
M
M
T
T
K
K
I
I
L
L
E
E
P
P
F
F
R
R
760
|
K
K
Q
Q
N
N
P
P
D
D
I
I
V
V
I
I
Y
Y
Q
Q
770
|
Y
Y
M
M
D
D
D
D
L
L
Y
Y
V
V
G
G
S
S
D
D
780
|
L
L
E
E
I
I
G
G
Q
Q
H
H
R
R
T
T
K
K
I
I
790
|
E
E
E
E
L
L
R
R
Q
Q
H
H
L
L
L
L
R
R
W
W
800
|
G
G
L
L
T
T
T
T
P
P
D
D
K
K
K
K
H
H
Q
Q
810
|
K
K
E
E
P
P
P
P
F
F
L
L
W
W
M
M
G
G
Y
Y
820
|
E
E
L
L
H
H
P
P
D
D
K
K
W
W
T
T
V
V
Q
Q
830
|
P
P
I
I
V
V
L
L
P
P
E
E
K
K
D
D
S
S
W
W
840
|
T
T
V
V
N
N
D
D
I
I
Q
Q
K
K
L
L
V
V
G
G
850
|
K
K
L
L
N
N
W
W
A
A
S
S
Q
Q
I
I
Y
Y
P
P
860
|
G
G
I
I
K
K
V
V
R
R
Q
Q
L
L
C
C
K
K
L
L
870
|
L
L
R
R
G
G
T
T
K
K
A
A
L
L
T
T
E
E
V
V
880
|
I
I
P
P
L
L
T
T
E
E
E
E
A
A
E
E
L
L
E
E
890
|
L
L
A
A
E
E
N
N
R
R
E
E
I
I
L
L
K
K
E
E
900
|
P
P
V
V
H
H
G
G
V
V
Y
Y
Y
Y
D
D
P
P
S
S
910
|
K
K
D
D
L
L
I
I
A
A
E
E
I
I
Q
Q
K
K
Q
Q
920
|
G
G
Q
Q
G
G
Q
Q
W
W
T
T
Y
Y
Q
Q
I
I
Y
Y
930
|
Q
Q
E
E
P
P
F
F
K
K
N
N
L
L
K
K
T
T
G
G
940
|
K
K
Y
Y
A
A
R
R
M
M
R
R
G
G
A
A
H
H
T
T
950
|
N
N
D
D
V
V
K
K
Q
Q
L
L
T
T
E
E
A
A
V
V
960
|
Q
Q
K
K
I
I
T
T
T
T
E
E
S
S
I
I
V
V
I
I
970
|
W
W
G
G
K
K
T
T
P
P
K
K
F
F
K
K
L
L
P
P
980
|
I
I
Q
Q
K
K
E
E
T
T
W
W
E
E
T
T
W
W
W
W
990
|
T
T
E
E
Y
Y
W
W
Q
Q
A
A
T
T
W
W
I
I
P
P
1000
|
E
E
W
W
E
E
F
F
V
V
N
N
T
T
P
P
P
P
L
L
1010
|
V
V
K
K
L
L
W
W
Y
Y
Q
Q
L
L
E
E
K
K
E
E
1020
|
P
P
I
I
V
V
G
G
A
A
E
E
T
T
F
F
Y
Y
V
V
1030
|
D
D
G
G
A
A
A
A
N
N
R
R
E
E
T
T
K
K
L
L
1040
|
G
G
K
K
A
A
G
G
Y
Y
V
V
T
T
N
N
R
R
G
G
1050
|
R
R
Q
Q
K
K
V
V
V
V
T
T
L
L
T
T
D
D
T
T
1060
|
T
T
N
N
Q
Q
K
K
T
T
E
E
L
L
Q
Q
A
A
I
I
1070
|
Y
Y
L
L
A
A
L
L
Q
Q
D
D
S
S
G
G
L
L
E
E
1080
|
V
V
N
N
I
I
V
V
T
T
D
D
S
S
Q
Q
Y
Y
A
A
1090
|
L
L
G
G
I
I
I
I
Q
Q
A
A
Q
Q
P
P
D
D
Q
Q
1100
|
S
S
E
E
S
S
E
E
L
L
V
V
N
N
Q
Q
I
I
I
I
1110
|
E
E
Q
Q
L
L
I
I
K
K
K
K
E
E
K
K
V
V
Y
Y
1120
|
L
L
A
A
W
W
V
V
P
P
A
A
H
H
K
K
G
G
I
I
1130
|
G
G
G
G
N
N
E
E
Q
Q
V
V
D
D
K
K
L
L
V
V
1140
|
S
S
A
A
G
G
I
I
R
R
K
K
V
V
L
L
|
|||||||||||||
| Experimental Note | Identified from the Human Clinical Data | ||||||||||||
| Experiment for Molecule Alteration |
Sanger sequencing assay; GeneSeq assay; Population sequencing of the integrase region assay; DNA sequencing assay; Site-directed mutagenesis | ||||||||||||
| Experiment for Drug Resistance |
PhenoSense assay; Viral load assay; A single-cycle assay; TZM-bl cell line-based phenotypic assay; SDMs and phenotyping assay | ||||||||||||
| Mechanism Description | Some mutations identified were assiacted with resistance in the HIV isolates, those mutations included D67N, T69N, K70R, V90I, K103N, V118I, I135M. | ||||||||||||
| Key Molecule: HIV1 Reverse transcriptase (HIV1 RT) | [1] | ||||||||||||
| Resistant Disease | Human immunodeficiency virus infection [ICD-11: 1C62.0] | ||||||||||||
| Molecule Alteration | Missense mutation | p.K101E+p.K102Q+p.S162C+p.K277R+p.I293V |
|||||||||||
| Experimental Note | Discovered Using In-vivo Testing Model | ||||||||||||
| In Vitro Model | HEK293T cells | Kidney | Homo sapiens (Human) | CVCL_0063 | |||||||||
| Experiment for Molecule Alteration |
Site-directed mutagenesis | ||||||||||||
| Experiment for Drug Resistance |
PhenoSense assay | ||||||||||||
| Mechanism Description | The k101E substitution conferred large reductions in susceptibility to NVP and DLV, while more modest reductions in susceptibility to EFV, ETR, or RPV were observed. In contrast, the E138k substitution did not confer large reductions in susceptibility to any of the NNRTIs and only small reductions in susceptibility to ETR and RPV. That is because that the k101E substitution compensates for the impaired viral replication capacity of HIV-1 containing M184I. | ||||||||||||
| Key Molecule: HIV1 Reverse transcriptase (HIV1 RT) | [1] | ||||||||||||
| Resistant Disease | Human immunodeficiency virus infection [ICD-11: 1C62.0] | ||||||||||||
| Molecule Alteration | Missense mutation | p.K102Q+p.E138K+p.S162C+p.K277R+p.I293V |
|||||||||||
| Experimental Note | Discovered Using In-vivo Testing Model | ||||||||||||
| In Vitro Model | HEK293T cells | Kidney | Homo sapiens (Human) | CVCL_0063 | |||||||||
| Experiment for Molecule Alteration |
Site-directed mutagenesis | ||||||||||||
| Experiment for Drug Resistance |
PhenoSense assay | ||||||||||||
| Mechanism Description | The k101E substitution conferred large reductions in susceptibility to NVP and DLV, while more modest reductions in susceptibility to EFV, ETR, or RPV were observed. In contrast, the E138k substitution did not confer large reductions in susceptibility to any of the NNRTIs and only small reductions in susceptibility to ETR and RPV. That is because that the k101E substitution compensates for the impaired viral replication capacity of HIV-1 containing M184I. | ||||||||||||
| Key Molecule: HIV1 Reverse transcriptase (HIV1 RT) | [1] | ||||||||||||
| Resistant Disease | Human immunodeficiency virus infection [ICD-11: 1C62.0] | ||||||||||||
| Molecule Alteration | Missense mutation | p.K101E+p.K102Q+p.E138K+p.S162C+p.M184I+p.K277R+p.I293V |
|||||||||||
| Experimental Note | Discovered Using In-vivo Testing Model | ||||||||||||
| In Vitro Model | HEK293T cells | Kidney | Homo sapiens (Human) | CVCL_0063 | |||||||||
| Experiment for Molecule Alteration |
Site-directed mutagenesis | ||||||||||||
| Experiment for Drug Resistance |
PhenoSense assay | ||||||||||||
| Mechanism Description | The k101E substitution conferred large reductions in susceptibility to NVP and DLV, while more modest reductions in susceptibility to EFV, ETR, or RPV were observed. In contrast, the E138k substitution did not confer large reductions in susceptibility to any of the NNRTIs and only small reductions in susceptibility to ETR and RPV. That is because that the k101E substitution compensates for the impaired viral replication capacity of HIV-1 containing M184I. | ||||||||||||
| Key Molecule: HIV1 Reverse transcriptase (HIV1 RT) | [1] | ||||||||||||
| Resistant Disease | Human immunodeficiency virus infection [ICD-11: 1C62.0] | ||||||||||||
| Molecule Alteration | Missense mutation | p.K102Q+p.E138K+p.S162C+p.M184I+p.K277R+p.I293V |
|||||||||||
| Experimental Note | Discovered Using In-vivo Testing Model | ||||||||||||
| In Vitro Model | HEK293T cells | Kidney | Homo sapiens (Human) | CVCL_0063 | |||||||||
| Experiment for Molecule Alteration |
Site-directed mutagenesis | ||||||||||||
| Experiment for Drug Resistance |
PhenoSense assay | ||||||||||||
| Mechanism Description | The k101E substitution conferred large reductions in susceptibility to NVP and DLV, while more modest reductions in susceptibility to EFV, ETR, or RPV were observed. In contrast, the E138k substitution did not confer large reductions in susceptibility to any of the NNRTIs and only small reductions in susceptibility to ETR and RPV. That is because that the k101E substitution compensates for the impaired viral replication capacity of HIV-1 containing M184I. | ||||||||||||
| Key Molecule: HIV1 Reverse transcriptase (HIV1 RT) | [1] | ||||||||||||
| Resistant Disease | Human immunodeficiency virus infection [ICD-11: 1C62.0] | ||||||||||||
| Molecule Alteration | Missense mutation | p.K101E+p.K102Q+p.E138K+p.S162C+p.K277R+p.I293V |
|||||||||||
| Experimental Note | Discovered Using In-vivo Testing Model | ||||||||||||
| In Vitro Model | HEK293T cells | Kidney | Homo sapiens (Human) | CVCL_0063 | |||||||||
| Experiment for Molecule Alteration |
Site-directed mutagenesis | ||||||||||||
| Experiment for Drug Resistance |
PhenoSense assay | ||||||||||||
| Mechanism Description | The k101E substitution conferred large reductions in susceptibility to NVP and DLV, while more modest reductions in susceptibility to EFV, ETR, or RPV were observed. In contrast, the E138k substitution did not confer large reductions in susceptibility to any of the NNRTIs and only small reductions in susceptibility to ETR and RPV. That is because that the k101E substitution compensates for the impaired viral replication capacity of HIV-1 containing M184I. | ||||||||||||
| Key Molecule: HIV1 Reverse transcriptase (HIV1 RT) | [3] | ||||||||||||
| Resistant Disease | Human immunodeficiency virus infection [ICD-11: 1C62.0] | ||||||||||||
| Molecule Alteration | Missense mutation | p.K20R+p.V35I+p.K101KE+p.V106VI+p.D123E+p.I135T+p.K173T+p.Q174K+p.D177E+p.Y181I+p.M184V+p.G196E+p.T200K+p.Q207A+p.P243T+p.V245K+p.D256E+p.A272P+p.E291D+p.V292I+p.I293V+p.E297A |
|||||||||||
| Experimental Note | Identified from the Human Clinical Data | ||||||||||||
| Experiment for Molecule Alteration |
GeneSeq assay; PhenoSense GT assay | ||||||||||||
| Experiment for Drug Resistance |
Viral load assay | ||||||||||||
| Mechanism Description | Amino acid changes in IN may contribute to drug resistance or sensitivity. | ||||||||||||
| Key Molecule: HIV1 Reverse transcriptase (HIV1 RT) | [3] | ||||||||||||
| Resistant Disease | Human immunodeficiency virus infection [ICD-11: 1C62.0] | ||||||||||||
| Molecule Alteration | Missense mutation | p.V35E+p.T39A+p.E138K+p.D177E+p.V179VI+p.Y181I+p.M184V+p.E194EK+p.G196EK+p.Q207E+p.R211K+p.V245K+p.I257L+p.K277R+p.T286A+p.I293V |
|||||||||||
| Experimental Note | Identified from the Human Clinical Data | ||||||||||||
| Experiment for Molecule Alteration |
GeneSeq assay; PhenoSense GT assay | ||||||||||||
| Experiment for Drug Resistance |
Viral load assay | ||||||||||||
| Mechanism Description | Amino acid changes in IN may contribute to drug resistance or sensitivity. | ||||||||||||
| Key Molecule: HIV1 Reverse transcriptase (HIV1 RT) | [3] | ||||||||||||
| Resistant Disease | Human immunodeficiency virus infection [ICD-11: 1C62.0] | ||||||||||||
| Molecule Alteration | Missense mutation | p.K20KR+p.A62AV+p.K65KN+p.V108VI+p.F171Y+p.D177E+p.I178IMV+p.M184MIV+p.T200A+p.Q207T+p.R211K+p.F227FC+p.L228LR+p.M230MI+p.L234LI+p.V245E+p.A272P |
|||||||||||
| Experimental Note | Identified from the Human Clinical Data | ||||||||||||
| Experiment for Molecule Alteration |
GeneSeq assay; PhenoSense GT assay | ||||||||||||
| Experiment for Drug Resistance |
Viral load assay | ||||||||||||
| Mechanism Description | Amino acid changes in IN may contribute to drug resistance or sensitivity. | ||||||||||||
| Key Molecule: HIV1 Reverse transcriptase (HIV1 RT) | [3] | ||||||||||||
| Resistant Disease | Human immunodeficiency virus infection [ICD-11: 1C62.0] | ||||||||||||
| Molecule Alteration | Missense mutation | p.V35IM+p.G45X+p.V90I+p.K101KE+p.K122E+p.D123N+p.I135L+p.S162C+p.Q174K+p.V179I+p.Y181I+p.M184V+p.G196E+p.A272P+p.A288T |
|||||||||||
| Experimental Note | Identified from the Human Clinical Data | ||||||||||||
| Experiment for Molecule Alteration |
GeneSeq assay; PhenoSense GT assay | ||||||||||||
| Experiment for Drug Resistance |
Viral load assay | ||||||||||||
| Mechanism Description | Amino acid changes in IN may contribute to drug resistance or sensitivity. | ||||||||||||
| Key Molecule: HIV1 Reverse transcriptase (HIV1 RT) | [3] | ||||||||||||
| Resistant Disease | Human immunodeficiency virus infection [ICD-11: 1C62.0] | ||||||||||||
| Molecule Alteration | Missense mutation | p.V35I+p.V90I+p.K122E+p.D123N+p.I135L+p.S162C+p.Q174K+p.V179I+p.Y181I+p.M184V+p.G196E+p.A272P+p.A288T |
|||||||||||
| Experimental Note | Identified from the Human Clinical Data | ||||||||||||
| Experiment for Molecule Alteration |
GeneSeq assay; PhenoSense GT assay | ||||||||||||
| Experiment for Drug Resistance |
Viral load assay | ||||||||||||
| Mechanism Description | Amino acid changes in IN may contribute to drug resistance or sensitivity. | ||||||||||||
| Key Molecule: HIV1 Reverse transcriptase (HIV1 RT) | [3] | ||||||||||||
| Resistant Disease | Human immunodeficiency virus infection [ICD-11: 1C62.0] | ||||||||||||
| Molecule Alteration | Missense mutation | p.K11T+p.V35I+p.E40K+p.K65R+p.T69T_X+p.I135L+p.D177E+p.I178M+p.V179VI+p.Y181C+p.Y188H+p.V189VI+p.G196E+p.K219KE+p.E291D+p.E297K |
|||||||||||
| Experimental Note | Identified from the Human Clinical Data | ||||||||||||
| Experiment for Molecule Alteration |
GeneSeq assay; PhenoSense GT assay | ||||||||||||
| Experiment for Drug Resistance |
Viral load assay | ||||||||||||
| Mechanism Description | Amino acid changes in IN may contribute to drug resistance or sensitivity. | ||||||||||||
| Key Molecule: HIV1 Reverse transcriptase (HIV1 RT) | [3] | ||||||||||||
| Resistant Disease | Human immunodeficiency virus infection [ICD-11: 1C62.0] | ||||||||||||
| Molecule Alteration | Missense mutation | p.V8I+p.K20R+p.A98AG+p.K101KE+p.K122KE+p.D123X+p.E138EK+p.I178IM+p.Y181YC+p.M184I+p.R211K+p.H221HY+p.V245I+p.E248ED+p.A272P+p.K277KR+p.I293V+p.P294S+p.E297R |
|||||||||||
| Experimental Note | Identified from the Human Clinical Data | ||||||||||||
| Experiment for Molecule Alteration |
GeneSeq assay; PhenoSense GT assay | ||||||||||||
| Experiment for Drug Resistance |
Viral load assay | ||||||||||||
| Mechanism Description | Amino acid changes in IN may contribute to drug resistance or sensitivity. | ||||||||||||
| Key Molecule: HIV1 Reverse transcriptase (HIV1 RT) | [3] | ||||||||||||
| Resistant Disease | Human immunodeficiency virus infection [ICD-11: 1C62.0] | ||||||||||||
| Molecule Alteration | Missense mutation | p.V35I+p.D67G+p.A98S+p.K101E+p.K122E+p.D123G+p.Y181C+p.M184I+p.T200A+p.K219E+p.M230L+p.V245E+p.K277R+p.I293V+p.P294A+p.E297K |
|||||||||||
| Experimental Note | Identified from the Human Clinical Data | ||||||||||||
| Experiment for Molecule Alteration |
GeneSeq assay; PhenoSense GT assay | ||||||||||||
| Experiment for Drug Resistance |
Viral load assay | ||||||||||||
| Mechanism Description | Amino acid changes in IN may contribute to drug resistance or sensitivity. | ||||||||||||
| Key Molecule: HIV1 Reverse transcriptase (HIV1 RT) | [3] | ||||||||||||
| Resistant Disease | Human immunodeficiency virus infection [ICD-11: 1C62.0] | ||||||||||||
| Molecule Alteration | Missense mutation | p.K20R+p.V35L+p.T39A+p.K122E+p.D123DN+p.I135R+p.K173E+p.Q174QR+p.D177E+p.V179I+p.Y181C+p.M184V+p.G196E+p.T200I+p.I202IV+p.R211K+p.K249KR+p.A288S+p.I293V |
|||||||||||
| Experimental Note | Identified from the Human Clinical Data | ||||||||||||
| Experiment for Molecule Alteration |
GeneSeq assay; PhenoSense GT assay | ||||||||||||
| Experiment for Drug Resistance |
Viral load assay | ||||||||||||
| Mechanism Description | Amino acid changes in IN may contribute to drug resistance or sensitivity. | ||||||||||||
| Key Molecule: HIV1 Reverse transcriptase (HIV1 RT) | [2] | ||||||||||||
| Resistant Disease | Human immunodeficiency virus infection [ICD-11: 1C62.0] | ||||||||||||
| Molecule Alteration | Missense mutation | p.P4S+p.D67DN+p.T69N+p.K70KR+p.V90I+p.L100LI+p.K122E+p.D123N+p.I135IL+p.S162C+p.D177E+p.V179I+p.M184I+p.Y188L+p.T200A+p.R211K+p.K219E+p.M230MIL+p.L234LI+p.A272P+p.K277R+p.T286A+p.E297K |
|||||||||||
| Experimental Note | Identified from the Human Clinical Data | ||||||||||||
| Experiment for Molecule Alteration |
Site-directed mutagenesis; Sanger sequencing assay | ||||||||||||
| Experiment for Drug Resistance |
PhenoSense assay | ||||||||||||
| Mechanism Description | Sixteen mutations at eight positions had a mean regression coefficient >=1.0 log10 (a contribution to decreased susceptibility of 10-fold or greater) for nevirapine, efavirenz, etravirine and/or rilpivirine: L100I, k101P, k103N/S, V106A/M, Y181C/I/V, Y188C/L, G190A/E/Q/S and F227C. With the exception of L100I and F227C, each mutation had a mean regression coefficient >=1.0 log10 for nevirapine. With the exception of k103S, V106A, Y181C/I/V and F227C, each had a mean regression coefficient >=1.0 log10 for efavirenz. Five mutations, k101P, Y181I/V, G190E and F227C, had a mean regression coefficient >=1.0 log10 for etravirine. Six mutations, L100I, k101P, Y181I/V, G190E and F227C, had a mean regression coefficient >= 1.0 log10 for rilpivirine. | ||||||||||||
| Key Molecule: HIV1 Reverse transcriptase (HIV1 RT) | [2] | ||||||||||||
| Resistant Disease | Human immunodeficiency virus infection [ICD-11: 1C62.0] | ||||||||||||
| Molecule Alteration | Missense mutation | p.P4T+p.E6D+p.K20R+p.A33G+p.V60I+p.D67N+p.T69N+p.K70R+p.L74I+p.K101E+p.K103S+p.V118I+p.D123E+p.I135T+p.E138A+p.M184V+p.G190A+p.D192N+p.I195KR+p.G196E+p.E203K+p.R211A+p.F214L+p.K219Q+p.H221Y+p.I257L+p.A272P+p.K281R+p.E291D+p.E297K |
|||||||||||
| Experimental Note | Identified from the Human Clinical Data | ||||||||||||
| Experiment for Molecule Alteration |
Site-directed mutagenesis; Sanger sequencing assay | ||||||||||||
| Experiment for Drug Resistance |
PhenoSense assay | ||||||||||||
| Mechanism Description | Sixteen mutations at eight positions had a mean regression coefficient >=1.0 log10 (a contribution to decreased susceptibility of 10-fold or greater) for nevirapine, efavirenz, etravirine and/or rilpivirine: L100I, k101P, k103N/S, V106A/M, Y181C/I/V, Y188C/L, G190A/E/Q/S and F227C. With the exception of L100I and F227C, each mutation had a mean regression coefficient >=1.0 log10 for nevirapine. With the exception of k103S, V106A, Y181C/I/V and F227C, each had a mean regression coefficient >=1.0 log10 for efavirenz. Five mutations, k101P, Y181I/V, G190E and F227C, had a mean regression coefficient >=1.0 log10 for etravirine. Six mutations, L100I, k101P, Y181I/V, G190E and F227C, had a mean regression coefficient >= 1.0 log10 for rilpivirine. | ||||||||||||
| Key Molecule: HIV1 Reverse transcriptase (HIV1 RT) | [2] | ||||||||||||
| Resistant Disease | Human immunodeficiency virus infection [ICD-11: 1C62.0] | ||||||||||||
| Molecule Alteration | Missense mutation | p.K20R+p.V35M+p.T69D+p.K102Q+p.K103N+p.K122E+p.E138G+p.S162SC+p.I178L+p.Q207E+p.A272P+p.K277R+p.L283I+p.I293V+p.E297A |
|||||||||||
| Experimental Note | Identified from the Human Clinical Data | ||||||||||||
| Experiment for Molecule Alteration |
Site-directed mutagenesis; Sanger sequencing assay | ||||||||||||
| Experiment for Drug Resistance |
PhenoSense assay | ||||||||||||
| Mechanism Description | Sixteen mutations at eight positions had a mean regression coefficient >=1.0 log10 (a contribution to decreased susceptibility of 10-fold or greater) for nevirapine, efavirenz, etravirine and/or rilpivirine: L100I, k101P, k103N/S, V106A/M, Y181C/I/V, Y188C/L, G190A/E/Q/S and F227C. With the exception of L100I and F227C, each mutation had a mean regression coefficient >=1.0 log10 for nevirapine. With the exception of k103S, V106A, Y181C/I/V and F227C, each had a mean regression coefficient >=1.0 log10 for efavirenz. Five mutations, k101P, Y181I/V, G190E and F227C, had a mean regression coefficient >=1.0 log10 for etravirine. Six mutations, L100I, k101P, Y181I/V, G190E and F227C, had a mean regression coefficient >= 1.0 log10 for rilpivirine. | ||||||||||||
| Key Molecule: HIV1 Reverse transcriptase (HIV1 RT) | [2] | ||||||||||||
| Resistant Disease | Human immunodeficiency virus infection [ICD-11: 1C62.0] | ||||||||||||
| Molecule Alteration | Missense mutation | p.K20R+p.V35L+p.M41L+p.K43E+p.D67N+p.T69N+p.K70R+p.K102Q+p.K103N+p.K122E+p.D123E+p.D177N+p.M184V+p.Y188L+p.G196E+p.I202V+p.R211T+p.T215F+p.D218E+p.K219Q+p.A272P+p.K277R+p.L283I+p.A288T+p.I293V+p.E297K+p.A304E |
|||||||||||
| Experimental Note | Identified from the Human Clinical Data | ||||||||||||
| Experiment for Molecule Alteration |
Site-directed mutagenesis; Sanger sequencing assay | ||||||||||||
| Experiment for Drug Resistance |
PhenoSense assay | ||||||||||||
| Mechanism Description | Sixteen mutations at eight positions had a mean regression coefficient >=1.0 log10 (a contribution to decreased susceptibility of 10-fold or greater) for nevirapine, efavirenz, etravirine and/or rilpivirine: L100I, k101P, k103N/S, V106A/M, Y181C/I/V, Y188C/L, G190A/E/Q/S and F227C. With the exception of L100I and F227C, each mutation had a mean regression coefficient >=1.0 log10 for nevirapine. With the exception of k103S, V106A, Y181C/I/V and F227C, each had a mean regression coefficient >=1.0 log10 for efavirenz. Five mutations, k101P, Y181I/V, G190E and F227C, had a mean regression coefficient >=1.0 log10 for etravirine. Six mutations, L100I, k101P, Y181I/V, G190E and F227C, had a mean regression coefficient >= 1.0 log10 for rilpivirine. | ||||||||||||
| Key Molecule: HIV1 Reverse transcriptase (HIV1 RT) | [2] | ||||||||||||
| Resistant Disease | Human immunodeficiency virus infection [ICD-11: 1C62.0] | ||||||||||||
| Molecule Alteration | Missense mutation | p.V35T+p.M41L+p.K43KN+p.V60I+p.K122E+p.K173KQ+p.D177EG+p.V179VI+p.Y188L+p.G196E+p.R211RK+p.T215D+p.V245E+p.I257L+p.K277R+p.I293V+p.E297R |
|||||||||||
| Experimental Note | Identified from the Human Clinical Data | ||||||||||||
| Experiment for Molecule Alteration |
Site-directed mutagenesis; Sanger sequencing assay | ||||||||||||
| Experiment for Drug Resistance |
PhenoSense assay | ||||||||||||
| Mechanism Description | Sixteen mutations at eight positions had a mean regression coefficient >=1.0 log10 (a contribution to decreased susceptibility of 10-fold or greater) for nevirapine, efavirenz, etravirine and/or rilpivirine: L100I, k101P, k103N/S, V106A/M, Y181C/I/V, Y188C/L, G190A/E/Q/S and F227C. With the exception of L100I and F227C, each mutation had a mean regression coefficient >=1.0 log10 for nevirapine. With the exception of k103S, V106A, Y181C/I/V and F227C, each had a mean regression coefficient >=1.0 log10 for efavirenz. Five mutations, k101P, Y181I/V, G190E and F227C, had a mean regression coefficient >=1.0 log10 for etravirine. Six mutations, L100I, k101P, Y181I/V, G190E and F227C, had a mean regression coefficient >= 1.0 log10 for rilpivirine. | ||||||||||||
| Key Molecule: HIV1 Reverse transcriptase (HIV1 RT) | [2] | ||||||||||||
| Resistant Disease | Human immunodeficiency virus infection [ICD-11: 1C62.0] | ||||||||||||
| Molecule Alteration | Missense mutation | p.V35T+p.M41L+p.L80I+p.K122E+p.D177E+p.Y188L+p.H198E+p.T215D+p.V245VE+p.I257IL+p.K277R+p.A288AT+p.I293V+p.E297R |
|||||||||||
| Experimental Note | Identified from the Human Clinical Data | ||||||||||||
| Experiment for Molecule Alteration |
Site-directed mutagenesis; Sanger sequencing assay | ||||||||||||
| Experiment for Drug Resistance |
PhenoSense assay | ||||||||||||
| Mechanism Description | Sixteen mutations at eight positions had a mean regression coefficient >=1.0 log10 (a contribution to decreased susceptibility of 10-fold or greater) for nevirapine, efavirenz, etravirine and/or rilpivirine: L100I, k101P, k103N/S, V106A/M, Y181C/I/V, Y188C/L, G190A/E/Q/S and F227C. With the exception of L100I and F227C, each mutation had a mean regression coefficient >=1.0 log10 for nevirapine. With the exception of k103S, V106A, Y181C/I/V and F227C, each had a mean regression coefficient >=1.0 log10 for efavirenz. Five mutations, k101P, Y181I/V, G190E and F227C, had a mean regression coefficient >=1.0 log10 for etravirine. Six mutations, L100I, k101P, Y181I/V, G190E and F227C, had a mean regression coefficient >= 1.0 log10 for rilpivirine. | ||||||||||||
| Key Molecule: HIV1 Reverse transcriptase (HIV1 RT) | [2] | ||||||||||||
| Resistant Disease | Human immunodeficiency virus infection [ICD-11: 1C62.0] | ||||||||||||
| Molecule Alteration | Missense mutation | p.T39AE+p.M41L+p.K43KR+p.E44D+p.L74LI+p.A98AG+p.K102KR+p.K103N+p.K122E+p.D123S+p.I135T+p.I142V+p.M184V+p.Y188L+p.H208Y+p.L210W+p.R211K+p.T215Y+p.L228H+p.R284K+p.T286A+p.P294T |
|||||||||||
| Experimental Note | Identified from the Human Clinical Data | ||||||||||||
| Experiment for Molecule Alteration |
Site-directed mutagenesis; Sanger sequencing assay | ||||||||||||
| Experiment for Drug Resistance |
PhenoSense assay | ||||||||||||
| Mechanism Description | Sixteen mutations at eight positions had a mean regression coefficient >=1.0 log10 (a contribution to decreased susceptibility of 10-fold or greater) for nevirapine, efavirenz, etravirine and/or rilpivirine: L100I, k101P, k103N/S, V106A/M, Y181C/I/V, Y188C/L, G190A/E/Q/S and F227C. With the exception of L100I and F227C, each mutation had a mean regression coefficient >=1.0 log10 for nevirapine. With the exception of k103S, V106A, Y181C/I/V and F227C, each had a mean regression coefficient >=1.0 log10 for efavirenz. Five mutations, k101P, Y181I/V, G190E and F227C, had a mean regression coefficient >=1.0 log10 for etravirine. Six mutations, L100I, k101P, Y181I/V, G190E and F227C, had a mean regression coefficient >= 1.0 log10 for rilpivirine. | ||||||||||||
| Key Molecule: HIV1 Reverse transcriptase (HIV1 RT) | [2] | ||||||||||||
| Resistant Disease | Human immunodeficiency virus infection [ICD-11: 1C62.0] | ||||||||||||
| Molecule Alteration | Missense mutation | p.T69TN+p.K101P+p.K103N+p.I135V+p.I142V+p.Q145V+p.M184V+p.T200A+p.V245N+p.E248D+p.A272P+p.K275R+p.I293IV |
|||||||||||
| Experimental Note | Identified from the Human Clinical Data | ||||||||||||
| Experiment for Molecule Alteration |
Site-directed mutagenesis; Sanger sequencing assay | ||||||||||||
| Experiment for Drug Resistance |
PhenoSense assay | ||||||||||||
| Mechanism Description | Sixteen mutations at eight positions had a mean regression coefficient >=1.0 log10 (a contribution to decreased susceptibility of 10-fold or greater) for nevirapine, efavirenz, etravirine and/or rilpivirine: L100I, k101P, k103N/S, V106A/M, Y181C/I/V, Y188C/L, G190A/E/Q/S and F227C. With the exception of L100I and F227C, each mutation had a mean regression coefficient >=1.0 log10 for nevirapine. With the exception of k103S, V106A, Y181C/I/V and F227C, each had a mean regression coefficient >=1.0 log10 for efavirenz. Five mutations, k101P, Y181I/V, G190E and F227C, had a mean regression coefficient >=1.0 log10 for etravirine. Six mutations, L100I, k101P, Y181I/V, G190E and F227C, had a mean regression coefficient >= 1.0 log10 for rilpivirine. | ||||||||||||
| Key Molecule: HIV1 Reverse transcriptase (HIV1 RT) | [2] | ||||||||||||
| Resistant Disease | Human immunodeficiency virus infection [ICD-11: 1C62.0] | ||||||||||||
| Molecule Alteration | Missense mutation | p.K22R+p.V35M+p.S162C+p.I178L+p.V179I+p.Y181C+p.M184V+p.Y188L+p.T200A+p.Q207E+p.V245E+p.A272P+p.K277R |
|||||||||||
| Experimental Note | Identified from the Human Clinical Data | ||||||||||||
| Experiment for Molecule Alteration |
Site-directed mutagenesis; Sanger sequencing assay | ||||||||||||
| Experiment for Drug Resistance |
PhenoSense assay | ||||||||||||
| Mechanism Description | Sixteen mutations at eight positions had a mean regression coefficient >=1.0 log10 (a contribution to decreased susceptibility of 10-fold or greater) for nevirapine, efavirenz, etravirine and/or rilpivirine: L100I, k101P, k103N/S, V106A/M, Y181C/I/V, Y188C/L, G190A/E/Q/S and F227C. With the exception of L100I and F227C, each mutation had a mean regression coefficient >=1.0 log10 for nevirapine. With the exception of k103S, V106A, Y181C/I/V and F227C, each had a mean regression coefficient >=1.0 log10 for efavirenz. Five mutations, k101P, Y181I/V, G190E and F227C, had a mean regression coefficient >=1.0 log10 for etravirine. Six mutations, L100I, k101P, Y181I/V, G190E and F227C, had a mean regression coefficient >= 1.0 log10 for rilpivirine. | ||||||||||||
| Key Molecule: HIV1 Reverse transcriptase (HIV1 RT) | [2] | ||||||||||||
| Resistant Disease | Human immunodeficiency virus infection [ICD-11: 1C62.0] | ||||||||||||
| Molecule Alteration | Missense mutation | p.S3C+p.E28R+p.K32E+p.V35M+p.M41L+p.L74I+p.R83K+p.A98G+p.K101E+p.K103N+p.V108I+p.S162A+p.I167IV+p.E169D+p.D177E+p.I178L+p.M184V+p.G196E+p.T200A+p.E203D+p.Q207E+p.L210W+p.R211K+p.T215Y+p.L228H+p.L260V+p.A272P+p.K281R+p.T286A+p.E297K |
|||||||||||
| Experimental Note | Identified from the Human Clinical Data | ||||||||||||
| Experiment for Molecule Alteration |
Site-directed mutagenesis; Sanger sequencing assay | ||||||||||||
| Experiment for Drug Resistance |
PhenoSense assay | ||||||||||||
| Mechanism Description | Sixteen mutations at eight positions had a mean regression coefficient >=1.0 log10 (a contribution to decreased susceptibility of 10-fold or greater) for nevirapine, efavirenz, etravirine and/or rilpivirine: L100I, k101P, k103N/S, V106A/M, Y181C/I/V, Y188C/L, G190A/E/Q/S and F227C. With the exception of L100I and F227C, each mutation had a mean regression coefficient >=1.0 log10 for nevirapine. With the exception of k103S, V106A, Y181C/I/V and F227C, each had a mean regression coefficient >=1.0 log10 for efavirenz. Five mutations, k101P, Y181I/V, G190E and F227C, had a mean regression coefficient >=1.0 log10 for etravirine. Six mutations, L100I, k101P, Y181I/V, G190E and F227C, had a mean regression coefficient >= 1.0 log10 for rilpivirine. | ||||||||||||
| Key Molecule: HIV1 Reverse transcriptase (HIV1 RT) | [2] | ||||||||||||
| Resistant Disease | Human immunodeficiency virus infection [ICD-11: 1C62.0] | ||||||||||||
| Molecule Alteration | Missense mutation | p.E28K+p.K32E+p.T39K+p.M41L+p.K43E+p.E44A+p.V60I+p.D67N+p.V75M+p.R83K+p.K102KR+p.K103S+p.V118I+p.K122E+p.I135T+p.V179T+p.Y181V+p.M184V+p.T200A+p.E203V+p.Q207E+p.H208Y+p.L210W+p.R211K+p.T215Y+p.D218E+p.K219E+p.L228H+p.Q242H+p.V245E+p.D256E+p.A272P+p.T286TI+p.A288T+p.I293V+p.E297T |
|||||||||||
| Experimental Note | Identified from the Human Clinical Data | ||||||||||||
| Experiment for Molecule Alteration |
Site-directed mutagenesis; Sanger sequencing assay | ||||||||||||
| Experiment for Drug Resistance |
PhenoSense assay | ||||||||||||
| Mechanism Description | Sixteen mutations at eight positions had a mean regression coefficient >=1.0 log10 (a contribution to decreased susceptibility of 10-fold or greater) for nevirapine, efavirenz, etravirine and/or rilpivirine: L100I, k101P, k103N/S, V106A/M, Y181C/I/V, Y188C/L, G190A/E/Q/S and F227C. With the exception of L100I and F227C, each mutation had a mean regression coefficient >=1.0 log10 for nevirapine. With the exception of k103S, V106A, Y181C/I/V and F227C, each had a mean regression coefficient >=1.0 log10 for efavirenz. Five mutations, k101P, Y181I/V, G190E and F227C, had a mean regression coefficient >=1.0 log10 for etravirine. Six mutations, L100I, k101P, Y181I/V, G190E and F227C, had a mean regression coefficient >= 1.0 log10 for rilpivirine. | ||||||||||||
| Key Molecule: HIV1 Reverse transcriptase (HIV1 RT) | [2] | ||||||||||||
| Resistant Disease | Human immunodeficiency virus infection [ICD-11: 1C62.0] | ||||||||||||
| Molecule Alteration | Missense mutation | p.I5IV+p.E6K+p.K20KR+p.E44ED+p.D67N+p.T69D+p.V90VI+p.K102Q+p.K103N+p.K104KR+p.V118I+p.K122E+p.I142IV+p.V179I+p.Y181C+p.Y188L+p.T200A+p.Q207N+p.R211K+p.F214L+p.H221Y+p.I293V |
|||||||||||
| Experimental Note | Identified from the Human Clinical Data | ||||||||||||
| Experiment for Molecule Alteration |
Site-directed mutagenesis; Sanger sequencing assay | ||||||||||||
| Experiment for Drug Resistance |
PhenoSense assay | ||||||||||||
| Mechanism Description | Sixteen mutations at eight positions had a mean regression coefficient >=1.0 log10 (a contribution to decreased susceptibility of 10-fold or greater) for nevirapine, efavirenz, etravirine and/or rilpivirine: L100I, k101P, k103N/S, V106A/M, Y181C/I/V, Y188C/L, G190A/E/Q/S and F227C. With the exception of L100I and F227C, each mutation had a mean regression coefficient >=1.0 log10 for nevirapine. With the exception of k103S, V106A, Y181C/I/V and F227C, each had a mean regression coefficient >=1.0 log10 for efavirenz. Five mutations, k101P, Y181I/V, G190E and F227C, had a mean regression coefficient >=1.0 log10 for etravirine. Six mutations, L100I, k101P, Y181I/V, G190E and F227C, had a mean regression coefficient >= 1.0 log10 for rilpivirine. | ||||||||||||
| Key Molecule: HIV1 Reverse transcriptase (HIV1 RT) | [2] | ||||||||||||
| Resistant Disease | Human immunodeficiency virus infection [ICD-11: 1C62.0] | ||||||||||||
| Molecule Alteration | Missense mutation | p.V35VI+p.T39TA+p.V90VI+p.L100I+p.Y115F+p.K122E+p.A158S+p.E169D+p.V179VI+p.M184V+p.Y188L+p.G196D+p.T200TA+p.Q207K+p.R211RK+p.T215F+p.K219E+p.L228H+p.V245E |
|||||||||||
| Experimental Note | Identified from the Human Clinical Data | ||||||||||||
| Experiment for Molecule Alteration |
Site-directed mutagenesis; Sanger sequencing assay | ||||||||||||
| Experiment for Drug Resistance |
PhenoSense assay | ||||||||||||
| Mechanism Description | Sixteen mutations at eight positions had a mean regression coefficient >=1.0 log10 (a contribution to decreased susceptibility of 10-fold or greater) for nevirapine, efavirenz, etravirine and/or rilpivirine: L100I, k101P, k103N/S, V106A/M, Y181C/I/V, Y188C/L, G190A/E/Q/S and F227C. With the exception of L100I and F227C, each mutation had a mean regression coefficient >=1.0 log10 for nevirapine. With the exception of k103S, V106A, Y181C/I/V and F227C, each had a mean regression coefficient >=1.0 log10 for efavirenz. Five mutations, k101P, Y181I/V, G190E and F227C, had a mean regression coefficient >=1.0 log10 for etravirine. Six mutations, L100I, k101P, Y181I/V, G190E and F227C, had a mean regression coefficient >= 1.0 log10 for rilpivirine. | ||||||||||||
| Key Molecule: HIV1 Reverse transcriptase (HIV1 RT) | [2] | ||||||||||||
| Resistant Disease | Human immunodeficiency virus infection [ICD-11: 1C62.0] | ||||||||||||
| Molecule Alteration | Missense mutation | p.M41L+p.K43Q+p.V60I+p.D67N+p.T69D+p.L74LV+p.V75VIM+p.L100I+p.K103N+p.K122KE+p.I135L+p.E138G+p.I142V+p.I178L+p.M184V+p.G196E+p.Q207E+p.T215Y+p.V245E+p.A272P+p.V276VI+p.K277KR+p.T286A+p.E291A+p.I293V |
|||||||||||
| Experimental Note | Identified from the Human Clinical Data | ||||||||||||
| Experiment for Molecule Alteration |
Site-directed mutagenesis; Sanger sequencing assay | ||||||||||||
| Experiment for Drug Resistance |
PhenoSense assay | ||||||||||||
| Mechanism Description | Sixteen mutations at eight positions had a mean regression coefficient >=1.0 log10 (a contribution to decreased susceptibility of 10-fold or greater) for nevirapine, efavirenz, etravirine and/or rilpivirine: L100I, k101P, k103N/S, V106A/M, Y181C/I/V, Y188C/L, G190A/E/Q/S and F227C. With the exception of L100I and F227C, each mutation had a mean regression coefficient >=1.0 log10 for nevirapine. With the exception of k103S, V106A, Y181C/I/V and F227C, each had a mean regression coefficient >=1.0 log10 for efavirenz. Five mutations, k101P, Y181I/V, G190E and F227C, had a mean regression coefficient >=1.0 log10 for etravirine. Six mutations, L100I, k101P, Y181I/V, G190E and F227C, had a mean regression coefficient >= 1.0 log10 for rilpivirine. | ||||||||||||
| Key Molecule: HIV1 Reverse transcriptase (HIV1 RT) | [2] | ||||||||||||
| Resistant Disease | Human immunodeficiency virus infection [ICD-11: 1C62.0] | ||||||||||||
| Molecule Alteration | Missense mutation | p.P4S+p.V35VA+p.S48E+p.K101KE+p.K103KN+p.E169A+p.K173E+p.I178IM+p.I180IL+p.T200TA+p.V245VI+p.A272P |
|||||||||||
| Experimental Note | Identified from the Human Clinical Data | ||||||||||||
| Experiment for Molecule Alteration |
Site-directed mutagenesis; Sanger sequencing assay | ||||||||||||
| Experiment for Drug Resistance |
PhenoSense assay | ||||||||||||
| Mechanism Description | Sixteen mutations at eight positions had a mean regression coefficient >=1.0 log10 (a contribution to decreased susceptibility of 10-fold or greater) for nevirapine, efavirenz, etravirine and/or rilpivirine: L100I, k101P, k103N/S, V106A/M, Y181C/I/V, Y188C/L, G190A/E/Q/S and F227C. With the exception of L100I and F227C, each mutation had a mean regression coefficient >=1.0 log10 for nevirapine. With the exception of k103S, V106A, Y181C/I/V and F227C, each had a mean regression coefficient >=1.0 log10 for efavirenz. Five mutations, k101P, Y181I/V, G190E and F227C, had a mean regression coefficient >=1.0 log10 for etravirine. Six mutations, L100I, k101P, Y181I/V, G190E and F227C, had a mean regression coefficient >= 1.0 log10 for rilpivirine. | ||||||||||||
| Key Molecule: HIV1 Reverse transcriptase (HIV1 RT) | [2] | ||||||||||||
| Resistant Disease | Human immunodeficiency virus infection [ICD-11: 1C62.0] | ||||||||||||
| Molecule Alteration | Missense mutation | p.V35L+p.D67N+p.K70R+p.K101R+p.K103R+p.D177E+p.I178M+p.G190E+p.G196E+p.T200A+p.E204N+p.R211K+p.T215F+p.K219E+p.L228H+p.V245E+p.A272P+p.T286A+p.E297K |
|||||||||||
| Experimental Note | Identified from the Human Clinical Data | ||||||||||||
| Experiment for Molecule Alteration |
Site-directed mutagenesis; Sanger sequencing assay | ||||||||||||
| Experiment for Drug Resistance |
PhenoSense assay | ||||||||||||
| Mechanism Description | Sixteen mutations at eight positions had a mean regression coefficient >=1.0 log10 (a contribution to decreased susceptibility of 10-fold or greater) for nevirapine, efavirenz, etravirine and/or rilpivirine: L100I, k101P, k103N/S, V106A/M, Y181C/I/V, Y188C/L, G190A/E/Q/S and F227C. With the exception of L100I and F227C, each mutation had a mean regression coefficient >=1.0 log10 for nevirapine. With the exception of k103S, V106A, Y181C/I/V and F227C, each had a mean regression coefficient >=1.0 log10 for efavirenz. Five mutations, k101P, Y181I/V, G190E and F227C, had a mean regression coefficient >=1.0 log10 for etravirine. Six mutations, L100I, k101P, Y181I/V, G190E and F227C, had a mean regression coefficient >= 1.0 log10 for rilpivirine. | ||||||||||||
| Key Molecule: HIV1 Reverse transcriptase (HIV1 RT) | [2] | ||||||||||||
| Resistant Disease | Human immunodeficiency virus infection [ICD-11: 1C62.0] | ||||||||||||
| Molecule Alteration | Missense mutation | p.D67N+p.K70R+p.L74IV+p.L100I+p.K103N+p.D123E+p.I135T+p.K166R+p.G196E+p.T200A+p.I202V+p.T215I+p.K219E+p.L228LR+p.K277R+p.L283I+p.A288T+p.I293V+p.P294I+p.E297P |
|||||||||||
| Experimental Note | Identified from the Human Clinical Data | ||||||||||||
| Experiment for Molecule Alteration |
Site-directed mutagenesis; Sanger sequencing assay | ||||||||||||
| Experiment for Drug Resistance |
PhenoSense assay | ||||||||||||
| Mechanism Description | Sixteen mutations at eight positions had a mean regression coefficient >=1.0 log10 (a contribution to decreased susceptibility of 10-fold or greater) for nevirapine, efavirenz, etravirine and/or rilpivirine: L100I, k101P, k103N/S, V106A/M, Y181C/I/V, Y188C/L, G190A/E/Q/S and F227C. With the exception of L100I and F227C, each mutation had a mean regression coefficient >=1.0 log10 for nevirapine. With the exception of k103S, V106A, Y181C/I/V and F227C, each had a mean regression coefficient >=1.0 log10 for efavirenz. Five mutations, k101P, Y181I/V, G190E and F227C, had a mean regression coefficient >=1.0 log10 for etravirine. Six mutations, L100I, k101P, Y181I/V, G190E and F227C, had a mean regression coefficient >= 1.0 log10 for rilpivirine. | ||||||||||||
| Key Molecule: HIV1 Reverse transcriptase (HIV1 RT) | [2] | ||||||||||||
| Resistant Disease | Human immunodeficiency virus infection [ICD-11: 1C62.0] | ||||||||||||
| Molecule Alteration | Missense mutation | p.D67G+p.S68G+p.K70R+p.K101Q+p.K122E+p.V179I+p.Y181I+p.I195T+p.Q207E+p.R211K+p.F214L+p.K219Q+p.H221Y+p.L228H+p.V245E+p.A272P+p.K277R+p.T286A+p.I293V |
|||||||||||
| Experimental Note | Identified from the Human Clinical Data | ||||||||||||
| Experiment for Molecule Alteration |
Site-directed mutagenesis; Sanger sequencing assay | ||||||||||||
| Experiment for Drug Resistance |
PhenoSense assay | ||||||||||||
| Mechanism Description | Sixteen mutations at eight positions had a mean regression coefficient >=1.0 log10 (a contribution to decreased susceptibility of 10-fold or greater) for nevirapine, efavirenz, etravirine and/or rilpivirine: L100I, k101P, k103N/S, V106A/M, Y181C/I/V, Y188C/L, G190A/E/Q/S and F227C. With the exception of L100I and F227C, each mutation had a mean regression coefficient >=1.0 log10 for nevirapine. With the exception of k103S, V106A, Y181C/I/V and F227C, each had a mean regression coefficient >=1.0 log10 for efavirenz. Five mutations, k101P, Y181I/V, G190E and F227C, had a mean regression coefficient >=1.0 log10 for etravirine. Six mutations, L100I, k101P, Y181I/V, G190E and F227C, had a mean regression coefficient >= 1.0 log10 for rilpivirine. | ||||||||||||
| Key Molecule: HIV1 Reverse transcriptase (HIV1 RT) | [2] | ||||||||||||
| Resistant Disease | Human immunodeficiency virus infection [ICD-11: 1C62.0] | ||||||||||||
| Molecule Alteration | Missense mutation | p.E6D+p.K103N+p.L109LS+p.D123E+p.I135T+p.S162A+p.M184IV+p.T200I+p.E204Q+p.Q207E+p.F214L+p.M230L+p.I244V+p.I293V |
|||||||||||
| Experimental Note | Identified from the Human Clinical Data | ||||||||||||
| Experiment for Molecule Alteration |
Site-directed mutagenesis; Sanger sequencing assay | ||||||||||||
| Experiment for Drug Resistance |
PhenoSense assay | ||||||||||||
| Mechanism Description | Sixteen mutations at eight positions had a mean regression coefficient >=1.0 log10 (a contribution to decreased susceptibility of 10-fold or greater) for nevirapine, efavirenz, etravirine and/or rilpivirine: L100I, k101P, k103N/S, V106A/M, Y181C/I/V, Y188C/L, G190A/E/Q/S and F227C. With the exception of L100I and F227C, each mutation had a mean regression coefficient >=1.0 log10 for nevirapine. With the exception of k103S, V106A, Y181C/I/V and F227C, each had a mean regression coefficient >=1.0 log10 for efavirenz. Five mutations, k101P, Y181I/V, G190E and F227C, had a mean regression coefficient >=1.0 log10 for etravirine. Six mutations, L100I, k101P, Y181I/V, G190E and F227C, had a mean regression coefficient >= 1.0 log10 for rilpivirine. | ||||||||||||
| Key Molecule: HIV1 Reverse transcriptase (HIV1 RT) | [2] | ||||||||||||
| Resistant Disease | Human immunodeficiency virus infection [ICD-11: 1C62.0] | ||||||||||||
| Molecule Alteration | Missense mutation | p.K11KRT+p.V60VI+p.W88C+p.D121H+p.K122E+p.I135T+p.D177E+p.Y181C+p.R211K+p.H221Y+p.V245E+p.V254VI+p.A272P+p.A288G+p.E291D+p.I293IV+p.E297K |
|||||||||||
| Experimental Note | Identified from the Human Clinical Data | ||||||||||||
| Experiment for Molecule Alteration |
Site-directed mutagenesis; Sanger sequencing assay | ||||||||||||
| Experiment for Drug Resistance |
PhenoSense assay | ||||||||||||
| Mechanism Description | Sixteen mutations at eight positions had a mean regression coefficient >=1.0 log10 (a contribution to decreased susceptibility of 10-fold or greater) for nevirapine, efavirenz, etravirine and/or rilpivirine: L100I, k101P, k103N/S, V106A/M, Y181C/I/V, Y188C/L, G190A/E/Q/S and F227C. With the exception of L100I and F227C, each mutation had a mean regression coefficient >=1.0 log10 for nevirapine. With the exception of k103S, V106A, Y181C/I/V and F227C, each had a mean regression coefficient >=1.0 log10 for efavirenz. Five mutations, k101P, Y181I/V, G190E and F227C, had a mean regression coefficient >=1.0 log10 for etravirine. Six mutations, L100I, k101P, Y181I/V, G190E and F227C, had a mean regression coefficient >= 1.0 log10 for rilpivirine. | ||||||||||||
| Key Molecule: HIV1 Reverse transcriptase (HIV1 RT) | [2] | ||||||||||||
| Resistant Disease | Human immunodeficiency virus infection [ICD-11: 1C62.0] | ||||||||||||
| Molecule Alteration | Missense mutation | p.E6D+p.K122E+p.E138A+p.I142T+p.D177E+p.I178M+p.I195L+p.I202V+p.R211K+p.V245I+p.A272P+p.L283I+p.I293V+p.E297A |
|||||||||||
| Experimental Note | Identified from the Human Clinical Data | ||||||||||||
| Experiment for Molecule Alteration |
Site-directed mutagenesis; Sanger sequencing assay | ||||||||||||
| Experiment for Drug Resistance |
PhenoSense assay | ||||||||||||
| Mechanism Description | Sixteen mutations at eight positions had a mean regression coefficient >=1.0 log10 (a contribution to decreased susceptibility of 10-fold or greater) for nevirapine, efavirenz, etravirine and/or rilpivirine: L100I, k101P, k103N/S, V106A/M, Y181C/I/V, Y188C/L, G190A/E/Q/S and F227C. With the exception of L100I and F227C, each mutation had a mean regression coefficient >=1.0 log10 for nevirapine. With the exception of k103S, V106A, Y181C/I/V and F227C, each had a mean regression coefficient >=1.0 log10 for efavirenz. Five mutations, k101P, Y181I/V, G190E and F227C, had a mean regression coefficient >=1.0 log10 for etravirine. Six mutations, L100I, k101P, Y181I/V, G190E and F227C, had a mean regression coefficient >= 1.0 log10 for rilpivirine. | ||||||||||||
| Key Molecule: HIV1 Reverse transcriptase (HIV1 RT) | [2] | ||||||||||||
| Resistant Disease | Human immunodeficiency virus infection [ICD-11: 1C62.0] | ||||||||||||
| Molecule Alteration | Missense mutation | p.K20R+p.D67N+p.K70R+p.K122E+p.I135V+p.E138A+p.S162C+p.M184MV+p.G190E+p.T200A+p.F214M+p.T215F+p.K219Q+p.L228H+p.V245E |
|||||||||||
| Experimental Note | Identified from the Human Clinical Data | ||||||||||||
| Experiment for Molecule Alteration |
Site-directed mutagenesis; Sanger sequencing assay | ||||||||||||
| Experiment for Drug Resistance |
PhenoSense assay | ||||||||||||
| Mechanism Description | Sixteen mutations at eight positions had a mean regression coefficient >=1.0 log10 (a contribution to decreased susceptibility of 10-fold or greater) for nevirapine, efavirenz, etravirine and/or rilpivirine: L100I, k101P, k103N/S, V106A/M, Y181C/I/V, Y188C/L, G190A/E/Q/S and F227C. With the exception of L100I and F227C, each mutation had a mean regression coefficient >=1.0 log10 for nevirapine. With the exception of k103S, V106A, Y181C/I/V and F227C, each had a mean regression coefficient >=1.0 log10 for efavirenz. Five mutations, k101P, Y181I/V, G190E and F227C, had a mean regression coefficient >=1.0 log10 for etravirine. Six mutations, L100I, k101P, Y181I/V, G190E and F227C, had a mean regression coefficient >= 1.0 log10 for rilpivirine. | ||||||||||||
| Key Molecule: HIV1 Reverse transcriptase (HIV1 RT) | [2] | ||||||||||||
| Resistant Disease | Human immunodeficiency virus infection [ICD-11: 1C62.0] | ||||||||||||
| Molecule Alteration | Missense mutation | p.K20R+p.L74LV+p.Y115F+p.K122E+p.Y181C+p.M184V+p.R211K+p.F214L+p.H221Y+p.V241VM+p.I293V+p.E297A |
|||||||||||
| Experimental Note | Identified from the Human Clinical Data | ||||||||||||
| Experiment for Molecule Alteration |
Site-directed mutagenesis; Sanger sequencing assay | ||||||||||||
| Experiment for Drug Resistance |
PhenoSense assay | ||||||||||||
| Mechanism Description | Sixteen mutations at eight positions had a mean regression coefficient >=1.0 log10 (a contribution to decreased susceptibility of 10-fold or greater) for nevirapine, efavirenz, etravirine and/or rilpivirine: L100I, k101P, k103N/S, V106A/M, Y181C/I/V, Y188C/L, G190A/E/Q/S and F227C. With the exception of L100I and F227C, each mutation had a mean regression coefficient >=1.0 log10 for nevirapine. With the exception of k103S, V106A, Y181C/I/V and F227C, each had a mean regression coefficient >=1.0 log10 for efavirenz. Five mutations, k101P, Y181I/V, G190E and F227C, had a mean regression coefficient >=1.0 log10 for etravirine. Six mutations, L100I, k101P, Y181I/V, G190E and F227C, had a mean regression coefficient >= 1.0 log10 for rilpivirine. | ||||||||||||
| Key Molecule: HIV1 Reverse transcriptase (HIV1 RT) | [2] | ||||||||||||
| Resistant Disease | Human immunodeficiency virus infection [ICD-11: 1C62.0] | ||||||||||||
| Molecule Alteration | Missense mutation | p.K11K*+p.K20R+p.K64R+p.D67G+p.K70R+p.K101P+p.K103N+p.D123E+p.R172K+p.M184V+p.Q207E+p.R211K+p.F214L+p.K219E+p.T253TS |
|||||||||||
| Experimental Note | Identified from the Human Clinical Data | ||||||||||||
| Experiment for Molecule Alteration |
Site-directed mutagenesis; Sanger sequencing assay | ||||||||||||
| Experiment for Drug Resistance |
PhenoSense assay | ||||||||||||
| Mechanism Description | Sixteen mutations at eight positions had a mean regression coefficient >=1.0 log10 (a contribution to decreased susceptibility of 10-fold or greater) for nevirapine, efavirenz, etravirine and/or rilpivirine: L100I, k101P, k103N/S, V106A/M, Y181C/I/V, Y188C/L, G190A/E/Q/S and F227C. With the exception of L100I and F227C, each mutation had a mean regression coefficient >=1.0 log10 for nevirapine. With the exception of k103S, V106A, Y181C/I/V and F227C, each had a mean regression coefficient >=1.0 log10 for efavirenz. Five mutations, k101P, Y181I/V, G190E and F227C, had a mean regression coefficient >=1.0 log10 for etravirine. Six mutations, L100I, k101P, Y181I/V, G190E and F227C, had a mean regression coefficient >= 1.0 log10 for rilpivirine. | ||||||||||||
| Key Molecule: HIV1 Reverse transcriptase (HIV1 RT) | [2] | ||||||||||||
| Resistant Disease | Human immunodeficiency virus infection [ICD-11: 1C62.0] | ||||||||||||
| Molecule Alteration | Missense mutation | p.T39TA+p.D67N+p.K70R+p.V90I+p.K101E+p.S162N+p.M184V+p.G190A+p.E203D+p.R211K+p.F214FL+p.K219H+p.E297A |
|||||||||||
| Experimental Note | Identified from the Human Clinical Data | ||||||||||||
| Experiment for Molecule Alteration |
Site-directed mutagenesis; Sanger sequencing assay | ||||||||||||
| Experiment for Drug Resistance |
PhenoSense assay | ||||||||||||
| Mechanism Description | Sixteen mutations at eight positions had a mean regression coefficient >=1.0 log10 (a contribution to decreased susceptibility of 10-fold or greater) for nevirapine, efavirenz, etravirine and/or rilpivirine: L100I, k101P, k103N/S, V106A/M, Y181C/I/V, Y188C/L, G190A/E/Q/S and F227C. With the exception of L100I and F227C, each mutation had a mean regression coefficient >=1.0 log10 for nevirapine. With the exception of k103S, V106A, Y181C/I/V and F227C, each had a mean regression coefficient >=1.0 log10 for efavirenz. Five mutations, k101P, Y181I/V, G190E and F227C, had a mean regression coefficient >=1.0 log10 for etravirine. Six mutations, L100I, k101P, Y181I/V, G190E and F227C, had a mean regression coefficient >= 1.0 log10 for rilpivirine. | ||||||||||||
| Key Molecule: HIV1 Reverse transcriptase (HIV1 RT) | [2] | ||||||||||||
| Resistant Disease | Human immunodeficiency virus infection [ICD-11: 1C62.0] | ||||||||||||
| Molecule Alteration | Missense mutation | p.M41L+p.E44D+p.D67N+p.V118VI+p.I135T+p.S162C+p.R172K+p.Y188L+p.V189I+p.T200A+p.L210W+p.T215Y+p.D218E+p.K219R+p.V245E+p.A272P |
|||||||||||
| Experimental Note | Identified from the Human Clinical Data | ||||||||||||
| Experiment for Molecule Alteration |
Site-directed mutagenesis; Sanger sequencing assay | ||||||||||||
| Experiment for Drug Resistance |
PhenoSense assay | ||||||||||||
| Mechanism Description | Sixteen mutations at eight positions had a mean regression coefficient >=1.0 log10 (a contribution to decreased susceptibility of 10-fold or greater) for nevirapine, efavirenz, etravirine and/or rilpivirine: L100I, k101P, k103N/S, V106A/M, Y181C/I/V, Y188C/L, G190A/E/Q/S and F227C. With the exception of L100I and F227C, each mutation had a mean regression coefficient >=1.0 log10 for nevirapine. With the exception of k103S, V106A, Y181C/I/V and F227C, each had a mean regression coefficient >=1.0 log10 for efavirenz. Five mutations, k101P, Y181I/V, G190E and F227C, had a mean regression coefficient >=1.0 log10 for etravirine. Six mutations, L100I, k101P, Y181I/V, G190E and F227C, had a mean regression coefficient >= 1.0 log10 for rilpivirine. | ||||||||||||
| Key Molecule: HIV1 Reverse transcriptase (HIV1 RT) | [2] | ||||||||||||
| Resistant Disease | Human immunodeficiency virus infection [ICD-11: 1C62.0] | ||||||||||||
| Molecule Alteration | Missense mutation | p.K70KR+p.K122E+p.A158S+p.E169D+p.M184V+p.Y188L+p.G196D+p.Q207K+p.T215F+p.V245E |
|||||||||||
| Experimental Note | Identified from the Human Clinical Data | ||||||||||||
| Experiment for Molecule Alteration |
Site-directed mutagenesis; Sanger sequencing assay | ||||||||||||
| Experiment for Drug Resistance |
PhenoSense assay | ||||||||||||
| Mechanism Description | Sixteen mutations at eight positions had a mean regression coefficient >=1.0 log10 (a contribution to decreased susceptibility of 10-fold or greater) for nevirapine, efavirenz, etravirine and/or rilpivirine: L100I, k101P, k103N/S, V106A/M, Y181C/I/V, Y188C/L, G190A/E/Q/S and F227C. With the exception of L100I and F227C, each mutation had a mean regression coefficient >=1.0 log10 for nevirapine. With the exception of k103S, V106A, Y181C/I/V and F227C, each had a mean regression coefficient >=1.0 log10 for efavirenz. Five mutations, k101P, Y181I/V, G190E and F227C, had a mean regression coefficient >=1.0 log10 for etravirine. Six mutations, L100I, k101P, Y181I/V, G190E and F227C, had a mean regression coefficient >= 1.0 log10 for rilpivirine. | ||||||||||||
| Key Molecule: HIV1 Reverse transcriptase (HIV1 RT) | [2] | ||||||||||||
| Resistant Disease | Human immunodeficiency virus infection [ICD-11: 1C62.0] | ||||||||||||
| Molecule Alteration | Missense mutation | p.K20R+p.V60I+p.T69N+p.K70R+p.I142V+p.V179D+p.Y188L+p.R211K+p.V245I+p.K277R+p.A288S+p.I293V+p.E297A |
|||||||||||
| Experimental Note | Identified from the Human Clinical Data | ||||||||||||
| Experiment for Molecule Alteration |
Site-directed mutagenesis; Sanger sequencing assay | ||||||||||||
| Experiment for Drug Resistance |
PhenoSense assay | ||||||||||||
| Mechanism Description | Sixteen mutations at eight positions had a mean regression coefficient >=1.0 log10 (a contribution to decreased susceptibility of 10-fold or greater) for nevirapine, efavirenz, etravirine and/or rilpivirine: L100I, k101P, k103N/S, V106A/M, Y181C/I/V, Y188C/L, G190A/E/Q/S and F227C. With the exception of L100I and F227C, each mutation had a mean regression coefficient >=1.0 log10 for nevirapine. With the exception of k103S, V106A, Y181C/I/V and F227C, each had a mean regression coefficient >=1.0 log10 for efavirenz. Five mutations, k101P, Y181I/V, G190E and F227C, had a mean regression coefficient >=1.0 log10 for etravirine. Six mutations, L100I, k101P, Y181I/V, G190E and F227C, had a mean regression coefficient >= 1.0 log10 for rilpivirine. | ||||||||||||
| Key Molecule: HIV1 Reverse transcriptase (HIV1 RT) | [2] | ||||||||||||
| Resistant Disease | Human immunodeficiency virus infection [ICD-11: 1C62.0] | ||||||||||||
| Molecule Alteration | Missense mutation | p.M16MV+p.M41L+p.L74V+p.L100I+p.K103N+p.K122E+p.I135T+p.T139TA+p.K173KR+p.I178L+p.R211K+p.T215Y+p.A272P+p.I293V+p.E297A |
|||||||||||
| Experimental Note | Identified from the Human Clinical Data | ||||||||||||
| Experiment for Molecule Alteration |
Site-directed mutagenesis; Sanger sequencing assay | ||||||||||||
| Experiment for Drug Resistance |
PhenoSense assay | ||||||||||||
| Mechanism Description | Sixteen mutations at eight positions had a mean regression coefficient >=1.0 log10 (a contribution to decreased susceptibility of 10-fold or greater) for nevirapine, efavirenz, etravirine and/or rilpivirine: L100I, k101P, k103N/S, V106A/M, Y181C/I/V, Y188C/L, G190A/E/Q/S and F227C. With the exception of L100I and F227C, each mutation had a mean regression coefficient >=1.0 log10 for nevirapine. With the exception of k103S, V106A, Y181C/I/V and F227C, each had a mean regression coefficient >=1.0 log10 for efavirenz. Five mutations, k101P, Y181I/V, G190E and F227C, had a mean regression coefficient >=1.0 log10 for etravirine. Six mutations, L100I, k101P, Y181I/V, G190E and F227C, had a mean regression coefficient >= 1.0 log10 for rilpivirine. | ||||||||||||
| Key Molecule: HIV1 Reverse transcriptase (HIV1 RT) | [2] | ||||||||||||
| Resistant Disease | Human immunodeficiency virus infection [ICD-11: 1C62.0] | ||||||||||||
| Molecule Alteration | Missense mutation | p.T7A+p.M41L+p.K43E+p.D67N+p.K101Q+p.V106I+p.D123E+p.I135L+p.Y181I+p.M184V+p.Q197L+p.L210W+p.R211K+p.T215Y+p.A272P+p.I293V+p.L303P |
|||||||||||
| Experimental Note | Identified from the Human Clinical Data | ||||||||||||
| Experiment for Molecule Alteration |
Site-directed mutagenesis; Sanger sequencing assay | ||||||||||||
| Experiment for Drug Resistance |
PhenoSense assay | ||||||||||||
| Mechanism Description | Sixteen mutations at eight positions had a mean regression coefficient >=1.0 log10 (a contribution to decreased susceptibility of 10-fold or greater) for nevirapine, efavirenz, etravirine and/or rilpivirine: L100I, k101P, k103N/S, V106A/M, Y181C/I/V, Y188C/L, G190A/E/Q/S and F227C. With the exception of L100I and F227C, each mutation had a mean regression coefficient >=1.0 log10 for nevirapine. With the exception of k103S, V106A, Y181C/I/V and F227C, each had a mean regression coefficient >=1.0 log10 for efavirenz. Five mutations, k101P, Y181I/V, G190E and F227C, had a mean regression coefficient >=1.0 log10 for etravirine. Six mutations, L100I, k101P, Y181I/V, G190E and F227C, had a mean regression coefficient >= 1.0 log10 for rilpivirine. | ||||||||||||
| Key Molecule: HIV1 Reverse transcriptase (HIV1 RT) | [2] | ||||||||||||
| Resistant Disease | Human immunodeficiency virus infection [ICD-11: 1C62.0] | ||||||||||||
| Molecule Alteration | Missense mutation | p.V60I+p.D67G+p.S68G+p.K70KR+p.L74V+p.I135V+p.E138A+p.K173E+p.I178M+p.G190E+p.R211A+p.K219N+p.V245E+p.E248D+p.D250E+p.A272P+p.T286A |
|||||||||||
| Experimental Note | Identified from the Human Clinical Data | ||||||||||||
| Experiment for Molecule Alteration |
Site-directed mutagenesis; Sanger sequencing assay | ||||||||||||
| Experiment for Drug Resistance |
PhenoSense assay | ||||||||||||
| Mechanism Description | Sixteen mutations at eight positions had a mean regression coefficient >=1.0 log10 (a contribution to decreased susceptibility of 10-fold or greater) for nevirapine, efavirenz, etravirine and/or rilpivirine: L100I, k101P, k103N/S, V106A/M, Y181C/I/V, Y188C/L, G190A/E/Q/S and F227C. With the exception of L100I and F227C, each mutation had a mean regression coefficient >=1.0 log10 for nevirapine. With the exception of k103S, V106A, Y181C/I/V and F227C, each had a mean regression coefficient >=1.0 log10 for efavirenz. Five mutations, k101P, Y181I/V, G190E and F227C, had a mean regression coefficient >=1.0 log10 for etravirine. Six mutations, L100I, k101P, Y181I/V, G190E and F227C, had a mean regression coefficient >= 1.0 log10 for rilpivirine. | ||||||||||||
| Key Molecule: HIV1 Reverse transcriptase (HIV1 RT) | [2] | ||||||||||||
| Resistant Disease | Human immunodeficiency virus infection [ICD-11: 1C62.0] | ||||||||||||
| Molecule Alteration | Missense mutation | p.V35K+p.M41L+p.V60I+p.D67N+p.T69D+p.K70R+p.L74V+p.K101E+p.K104N+p.V118I+p.K122E+p.I135T+p.S162D+p.Y181C+p.M184V+p.G190S+p.F214L+p.T215F+p.K219Q+p.A272P |
|||||||||||
| Experimental Note | Identified from the Human Clinical Data | ||||||||||||
| Experiment for Molecule Alteration |
Site-directed mutagenesis; Sanger sequencing assay | ||||||||||||
| Experiment for Drug Resistance |
PhenoSense assay | ||||||||||||
| Mechanism Description | Sixteen mutations at eight positions had a mean regression coefficient >=1.0 log10 (a contribution to decreased susceptibility of 10-fold or greater) for nevirapine, efavirenz, etravirine and/or rilpivirine: L100I, k101P, k103N/S, V106A/M, Y181C/I/V, Y188C/L, G190A/E/Q/S and F227C. With the exception of L100I and F227C, each mutation had a mean regression coefficient >=1.0 log10 for nevirapine. With the exception of k103S, V106A, Y181C/I/V and F227C, each had a mean regression coefficient >=1.0 log10 for efavirenz. Five mutations, k101P, Y181I/V, G190E and F227C, had a mean regression coefficient >=1.0 log10 for etravirine. Six mutations, L100I, k101P, Y181I/V, G190E and F227C, had a mean regression coefficient >= 1.0 log10 for rilpivirine. | ||||||||||||
| Key Molecule: HIV1 Reverse transcriptase (HIV1 RT) | [4] | ||||||||||||
| Resistant Disease | Human immunodeficiency virus infection [ICD-11: 1C62.0] | ||||||||||||
| Molecule Alteration | Missense mutation | p.K20KR+p.T165TI+p.T200AV+p.E204EG+p.F214FL+p.K277R+p.T286A+p.A288S |
|||||||||||
| Experimental Note | Identified from the Human Clinical Data | ||||||||||||
| Experiment for Molecule Alteration |
Sanger sequencing assay; GeneSeq assay; Population sequencing of the integrase region assay; DNA sequencing assay; Site-directed mutagenesis | ||||||||||||
| Experiment for Drug Resistance |
PhenoSense assay; Viral load assay; A single-cycle assay; TZM-bl cell line-based phenotypic assay; SDMs and phenotyping assay | ||||||||||||
| Mechanism Description | Some mutations identified were assiacted with resistance in the HIV isolates, those mutations included K20KR, T165TI, T200AV, E204EG, F214FL, K277R, T286A, A288S. | ||||||||||||
| Key Molecule: HIV1 Reverse transcriptase (HIV1 RT) | [7] | ||||||||||||
| Resistant Disease | Human immunodeficiency virus infection [ICD-11: 1C62.0] | ||||||||||||
| Molecule Alteration | Missense mutation | p.K122E+p.M184I+p.F214L+p.A272P+p.K277R+p.A376T+p.A400T |
|||||||||||
| Experimental Note | Identified from the Human Clinical Data | ||||||||||||
| Experiment for Molecule Alteration |
Sanger sequencing assay; GeneSeq assay; Population sequencing of the integrase region assay; DNA sequencing assay; Site-directed mutagenesis | ||||||||||||
| Experiment for Drug Resistance |
PhenoSense assay; Viral load assay; A single-cycle assay; TZM-bl cell line-based phenotypic assay; SDMs and phenotyping assay | ||||||||||||
| Mechanism Description | Some mutations identified were assiacted with resistance in the HIV isolates, those mutations included K122E, M184I, F214L, A272P, K277R, A376T, A400T. | ||||||||||||
| Key Molecule: HIV1 Reverse transcriptase (HIV1 RT) | [7] | ||||||||||||
| Resistant Disease | Human immunodeficiency virus infection [ICD-11: 1C62.0] | ||||||||||||
| Molecule Alteration | Missense mutation | p.K122E+p.F214L+p.A272P+p.K277R+p.A376T+p.A400T |
|||||||||||
| Experimental Note | Identified from the Human Clinical Data | ||||||||||||
| Experiment for Molecule Alteration |
Sanger sequencing assay; GeneSeq assay; Population sequencing of the integrase region assay; DNA sequencing assay; Site-directed mutagenesis | ||||||||||||
| Experiment for Drug Resistance |
PhenoSense assay; Viral load assay; A single-cycle assay; TZM-bl cell line-based phenotypic assay; SDMs and phenotyping assay | ||||||||||||
| Mechanism Description | Some mutations identified were assiacted with resistance in the HIV isolates, those mutations included K122E, F214L, A272P, K277R, A376T, A400T. | ||||||||||||
| Key Molecule: HIV1 Reverse transcriptase (HIV1 RT) | [4] | ||||||||||||
| Resistant Disease | Human immunodeficiency virus infection [ICD-11: 1C62.0] | ||||||||||||
| Molecule Alteration | Missense mutation | p.V35I+p.R83K+p.K103N+p.I135T+p.E138G+p.K166KR+p.T200TA+p.H208HY+p.R211K+p.I244V+p.D250E+p.A272P+p.I293V |
|||||||||||
| Experimental Note | Identified from the Human Clinical Data | ||||||||||||
| Experiment for Molecule Alteration |
Sanger sequencing assay; GeneSeq assay; Population sequencing of the integrase region assay; DNA sequencing assay; Site-directed mutagenesis | ||||||||||||
| Experiment for Drug Resistance |
PhenoSense assay; Viral load assay; A single-cycle assay; TZM-bl cell line-based phenotypic assay; SDMs and phenotyping assay | ||||||||||||
| Mechanism Description | Some mutations identified were assiacted with resistance in the HIV isolates, those mutations included V35I, R83K, K103N, I135T, E138G, K166KR, T200TA, H208HY, R211K, I244V, D250E, A272P, I293V. | ||||||||||||
| Key Molecule: HIV1 Reverse transcriptase (HIV1 RT) | [4] | ||||||||||||
| Resistant Disease | Human immunodeficiency virus infection [ICD-11: 1C62.0] | ||||||||||||
| Molecule Alteration | Missense mutation | p.V35IM+p.T39TA+p.R83K+p.K102KQ+p.K103N+p.D123E+p.S162C+p.D177E+p.I178R+p.V179F+p.R211RK+p.V245EQ+p.D250E+p.K277R+p.T286A+p.I293V+p.M357AS+p.E370ED+p.T377TMI |
|||||||||||
| Experimental Note | Identified from the Human Clinical Data | ||||||||||||
| Experiment for Molecule Alteration |
Sanger sequencing assay; GeneSeq assay; Population sequencing of the integrase region assay; DNA sequencing assay; Site-directed mutagenesis | ||||||||||||
| Experiment for Drug Resistance |
PhenoSense assay; Viral load assay; A single-cycle assay; TZM-bl cell line-based phenotypic assay; SDMs and phenotyping assay | ||||||||||||
| Mechanism Description | Some mutations identified were assiacted with resistance in the HIV isolates, those mutations included V35IM, T39TA, R83K, K102KQ, K103N, D123E, S162C, D177E, I178R, V179F, R211RK, V245EQ, D250E, K277R, T286A, I293V, M357AS, E370ED, T377TMI. | ||||||||||||
| Key Molecule: HIV1 Reverse transcriptase (HIV1 RT) | [4] | ||||||||||||
| Resistant Disease | Human immunodeficiency virus infection [ICD-11: 1C62.0] | ||||||||||||
| Molecule Alteration | Missense mutation | p.K20KR+p.K22R+p.V106I+p.I178L+p.R211K+p.F214FL+p.D250E+p.S251SN+p.K277R |
|||||||||||
| Experimental Note | Identified from the Human Clinical Data | ||||||||||||
| Experiment for Molecule Alteration |
Sanger sequencing assay; GeneSeq assay; Population sequencing of the integrase region assay; DNA sequencing assay; Site-directed mutagenesis | ||||||||||||
| Experiment for Drug Resistance |
PhenoSense assay; Viral load assay; A single-cycle assay; TZM-bl cell line-based phenotypic assay; SDMs and phenotyping assay | ||||||||||||
| Mechanism Description | Some mutations identified were assiacted with resistance in the HIV isolates, those mutations included K20KR, K22R, V106I, I178L, R211K, F214FL, D250E, S251SN, K277R. | ||||||||||||
| Key Molecule: HIV1 Reverse transcriptase (HIV1 RT) | [4] | ||||||||||||
| Resistant Disease | Human immunodeficiency virus infection [ICD-11: 1C62.0] | ||||||||||||
| Molecule Alteration | Missense mutation | p.K20KR+p.V35I+p.K101H+p.I135V+p.S162C+p.D177E+p.I178IV+p.G190A+p.T200E+p.E203ED+p.R211K+p.I244IV+p.V245M+p.A272P+p.T286A+p.I293V+p.E297A |
|||||||||||
| Experimental Note | Identified from the Human Clinical Data | ||||||||||||
| Experiment for Molecule Alteration |
Sanger sequencing assay; GeneSeq assay; Population sequencing of the integrase region assay; DNA sequencing assay; Site-directed mutagenesis | ||||||||||||
| Experiment for Drug Resistance |
PhenoSense assay; Viral load assay; A single-cycle assay; TZM-bl cell line-based phenotypic assay; SDMs and phenotyping assay | ||||||||||||
| Mechanism Description | Some mutations identified were assiacted with resistance in the HIV isolates, those mutations included K20KR, V35I, K101H, I135V, S162C, D177E, I178IV, G190A, T200E, E203ED, R211K, I244IV, V245M, A272P, T286A, I293V, E297A. | ||||||||||||
| Key Molecule: HIV1 Reverse transcriptase (HIV1 RT) | [4] | ||||||||||||
| Resistant Disease | Human immunodeficiency virus infection [ICD-11: 1C62.0] | ||||||||||||
| Molecule Alteration | Missense mutation | p.K20R+p.V35M+p.T69D+p.K102Q+p.K122KE+p.E138EAG+p.S162C+p.I178L+p.M184MV+p.Q207E+p.A272P+p.K277R+p.L283I+p.I293V+p.E297A |
|||||||||||
| Experimental Note | Identified from the Human Clinical Data | ||||||||||||
| Experiment for Molecule Alteration |
Sanger sequencing assay; GeneSeq assay; Population sequencing of the integrase region assay; DNA sequencing assay; Site-directed mutagenesis | ||||||||||||
| Experiment for Drug Resistance |
PhenoSense assay; Viral load assay; A single-cycle assay; TZM-bl cell line-based phenotypic assay; SDMs and phenotyping assay | ||||||||||||
| Mechanism Description | Some mutations identified were assiacted with resistance in the HIV isolates, those mutations included K20R, V35M, T69D, K102Q, K122KE, E138EAG, S162C, I178L, M184MV, Q207E, A272P, K277R, L283I, I293V, E297A. | ||||||||||||
| Key Molecule: HIV1 Reverse transcriptase (HIV1 RT) | [7] | ||||||||||||
| Resistant Disease | Human immunodeficiency virus infection [ICD-11: 1C62.0] | ||||||||||||
| Molecule Alteration | Missense mutation | p.K122E+p.E138K+p.M184V+p.F214L+p.A272P+p.K277R+p.A376T+p.A400T |
|||||||||||
| Experimental Note | Identified from the Human Clinical Data | ||||||||||||
| Experiment for Molecule Alteration |
Sanger sequencing assay; GeneSeq assay; Population sequencing of the integrase region assay; DNA sequencing assay; Site-directed mutagenesis | ||||||||||||
| Experiment for Drug Resistance |
PhenoSense assay; Viral load assay; A single-cycle assay; TZM-bl cell line-based phenotypic assay; SDMs and phenotyping assay | ||||||||||||
| Mechanism Description | Some mutations identified were assiacted with resistance in the HIV isolates, those mutations included K122E, E138K, M184V, F214L, A272P, K277R, A376T, A400T. | ||||||||||||
| Key Molecule: HIV1 Reverse transcriptase (HIV1 RT) | [4] | ||||||||||||
| Resistant Disease | Human immunodeficiency virus infection [ICD-11: 1C62.0] | ||||||||||||
| Molecule Alteration | Missense mutation | p.D67G+p.K70E+p.V90I+p.K122E+p.I135IT+p.S162F+p.I178IM+p.Y181C+p.T200E+p.R211HQ+p.V245E+p.A272P+p.K277R+p.T286A+p.E297K |
|||||||||||
| Experimental Note | Identified from the Human Clinical Data | ||||||||||||
| Experiment for Molecule Alteration |
Sanger sequencing assay; GeneSeq assay; Population sequencing of the integrase region assay; DNA sequencing assay; Site-directed mutagenesis | ||||||||||||
| Experiment for Drug Resistance |
PhenoSense assay; Viral load assay; A single-cycle assay; TZM-bl cell line-based phenotypic assay; SDMs and phenotyping assay | ||||||||||||
| Mechanism Description | Some mutations identified were assiacted with resistance in the HIV isolates, those mutations included D67G, K70E, V90I, K122E, I135IT, S162F, I178IM, Y181C, T200E, R211HQ, V245E, A272P, K277R, T286A, E297K. | ||||||||||||
| Key Molecule: HIV1 Reverse transcriptase (HIV1 RT) | [4] | ||||||||||||
| Resistant Disease | Human immunodeficiency virus infection [ICD-11: 1C62.0] | ||||||||||||
| Molecule Alteration | Missense mutation | p.D67G+p.K70E+p.V90I+p.K102Q+p.I135T+p.S162F+p.I178IM+p.Y181C+p.T200E+p.R211QH+p.V245E+p.K277R+p.T286A+p.I293V+p.E297K |
|||||||||||
| Experimental Note | Identified from the Human Clinical Data | ||||||||||||
| Experiment for Molecule Alteration |
Sanger sequencing assay; GeneSeq assay; Population sequencing of the integrase region assay; DNA sequencing assay; Site-directed mutagenesis | ||||||||||||
| Experiment for Drug Resistance |
PhenoSense assay; Viral load assay; A single-cycle assay; TZM-bl cell line-based phenotypic assay; SDMs and phenotyping assay | ||||||||||||
| Mechanism Description | Some mutations identified were assiacted with resistance in the HIV isolates, those mutations included D67G, K70E, V90I, K102Q, I135T, S162F, I178IM, Y181C, T200E, R211QH, V245E, K277R, T286A, I293V, E297K. | ||||||||||||
| Key Molecule: HIV1 Reverse transcriptase (HIV1 RT) | [7] | ||||||||||||
| Resistant Disease | Human immunodeficiency virus infection [ICD-11: 1C62.0] | ||||||||||||
| Molecule Alteration | Missense mutation | p.K122E+p.E138K+p.F214L+p.A272P+p.K277R+p.A376T+p.A400T |
|||||||||||
| Experimental Note | Identified from the Human Clinical Data | ||||||||||||
| Experiment for Molecule Alteration |
Sanger sequencing assay; GeneSeq assay; Population sequencing of the integrase region assay; DNA sequencing assay; Site-directed mutagenesis | ||||||||||||
| Experiment for Drug Resistance |
PhenoSense assay; Viral load assay; A single-cycle assay; TZM-bl cell line-based phenotypic assay; SDMs and phenotyping assay | ||||||||||||
| Mechanism Description | Some mutations identified were assiacted with resistance in the HIV isolates, those mutations included K122E, E138K, F214L, A272P, K277R, A376T, A400T. | ||||||||||||
| Key Molecule: HIV1 Reverse transcriptase (HIV1 RT) | [7] | ||||||||||||
| Resistant Disease | Human immunodeficiency virus infection [ICD-11: 1C62.0] | ||||||||||||
| Molecule Alteration | Missense mutation | p.K122E+p.E138K+p.M184I+p.F214L+p.A272P+p.K277R+p.A376T+p.A400T |
|||||||||||
| Experimental Note | Identified from the Human Clinical Data | ||||||||||||
| Experiment for Molecule Alteration |
Sanger sequencing assay; GeneSeq assay; Population sequencing of the integrase region assay; DNA sequencing assay; Site-directed mutagenesis | ||||||||||||
| Experiment for Drug Resistance |
PhenoSense assay; Viral load assay; A single-cycle assay; TZM-bl cell line-based phenotypic assay; SDMs and phenotyping assay | ||||||||||||
| Mechanism Description | Some mutations identified were assiacted with resistance in the HIV isolates, those mutations included K122E, E138K, M184I, F214L, A272P, K277R, A376T, A400T. | ||||||||||||
| Key Molecule: HIV1 Reverse transcriptase (HIV1 RT) | [4] | ||||||||||||
| Resistant Disease | Human immunodeficiency virus infection [ICD-11: 1C62.0] | ||||||||||||
| Molecule Alteration | Missense mutation | p.M41L+p.A62V+p.D67N+p.T69D+p.V75I+p.R83K+p.K103N+p.V108I+p.F116Y+p.D123E+p.Q151M+p.K166R+p.D177E+p.I202V+p.L210W+p.T215Y+p.M230L+p.A272P+p.I293V+p.E297K |
|||||||||||
| Experimental Note | Identified from the Human Clinical Data | ||||||||||||
| Experiment for Molecule Alteration |
Sanger sequencing assay; GeneSeq assay; Population sequencing of the integrase region assay; DNA sequencing assay; Site-directed mutagenesis | ||||||||||||
| Experiment for Drug Resistance |
PhenoSense assay; Viral load assay; A single-cycle assay; TZM-bl cell line-based phenotypic assay; SDMs and phenotyping assay | ||||||||||||
| Mechanism Description | Some mutations identified were assiacted with resistance in the HIV isolates, those mutations included M41L, A62V, D67N, T69D, V75I, R83K, K103N, V108I, F116Y, D123E, Q151M, K166R, D177E, I202V, L210W, T215Y, M230L, A272P, I293V, E297K. | ||||||||||||
| Key Molecule: HIV1 Reverse transcriptase (HIV1 RT) | [4] | ||||||||||||
| Resistant Disease | Human immunodeficiency virus infection [ICD-11: 1C62.0] | ||||||||||||
| Molecule Alteration | Missense mutation | p.D123E+p.E138EK+p.S162C+p.I178L+p.Q207E+p.R211K+p.A272P+p.K277R+p.T286A+p.A288T+p.E297K |
|||||||||||
| Experimental Note | Identified from the Human Clinical Data | ||||||||||||
| Experiment for Molecule Alteration |
Sanger sequencing assay; GeneSeq assay; Population sequencing of the integrase region assay; DNA sequencing assay; Site-directed mutagenesis | ||||||||||||
| Experiment for Drug Resistance |
PhenoSense assay; Viral load assay; A single-cycle assay; TZM-bl cell line-based phenotypic assay; SDMs and phenotyping assay | ||||||||||||
| Mechanism Description | Some mutations identified were assiacted with resistance in the HIV isolates, those mutations included D123E, E138EK, S162C, I178L, Q207E, R211K, A272P, K277R, T286A, A288T, E297K. | ||||||||||||
| Key Molecule: HIV1 Reverse transcriptase (HIV1 RT) | [4] | ||||||||||||
| Resistant Disease | Human immunodeficiency virus infection [ICD-11: 1C62.0] | ||||||||||||
| Molecule Alteration | Missense mutation | p.V35M+p.K70E+p.K102Q+p.K103N+p.D123E+p.S162C+p.K173E+p.Q174R+p.I178L+p.M184V+p.T200A+p.R211K+p.L228HR+p.M230L+p.L234I+p.V245KQ+p.K277R+p.L283I+p.V292I+p.I293V+p.Q334QR+p.G335X+p.T369TA+p.V381VI |
|||||||||||
| Experimental Note | Identified from the Human Clinical Data | ||||||||||||
| Experiment for Molecule Alteration |
Sanger sequencing assay; GeneSeq assay; Population sequencing of the integrase region assay; DNA sequencing assay; Site-directed mutagenesis | ||||||||||||
| Experiment for Drug Resistance |
PhenoSense assay; Viral load assay; A single-cycle assay; TZM-bl cell line-based phenotypic assay; SDMs and phenotyping assay | ||||||||||||
| Mechanism Description | Some mutations identified were assiacted with resistance in the HIV isolates, those mutations included V35M, K70E, K102Q, K103N, D123E, S162C, K173E, Q174R, I178L, M184V, T200A, R211K, L228HR, M230L, L234I, V245KQ, K277R, L283I, V292I, I293V, Q334QR, G335X, T369TA, V381VI. | ||||||||||||
| Key Molecule: HIV1 Reverse transcriptase (HIV1 RT) | [8] | ||||||||||||
| Resistant Disease | Human immunodeficiency virus infection [ICD-11: 1C62.0] | ||||||||||||
| Molecule Alteration | Missense mutation | p.M41L+p.A98G+p.K101E+p.K122E+p.I135T+p.E138K+p.S162Y+p.Y181C+p.T200A+p.L210F+p.T215D+p.P243T+p.V245E+p.D250E+p.A272P+p.I274V+p.Q278H+p.K281R+p.T286A+p.A288S+p.K311R |
|||||||||||
| Experimental Note | Identified from the Human Clinical Data | ||||||||||||
| Experiment for Molecule Alteration |
Sanger sequencing assay; GeneSeq assay; Population sequencing of the integrase region assay; DNA sequencing assay; Site-directed mutagenesis | ||||||||||||
| Experiment for Drug Resistance |
PhenoSense assay; Viral load assay; A single-cycle assay; TZM-bl cell line-based phenotypic assay; SDMs and phenotyping assay | ||||||||||||
| Mechanism Description | Some mutations identified were assiacted with resistance in the HIV isolates, those mutations included M41L, A98G, K101E, K122E, I135T, E138K, S162Y, Y181C, T200A, L210F, T215D, P243T, V245E, D250E, A272P, I274V, Q278H, K281R, T286A, A288S, K311R. | ||||||||||||
| Key Molecule: HIV1 Reverse transcriptase (HIV1 RT) | [8] | ||||||||||||
| Resistant Disease | Human immunodeficiency virus infection [ICD-11: 1C62.0] | ||||||||||||
| Molecule Alteration | Missense mutation | p.M41L+p.A98S+p.K101P+p.K102Q+p.K103N+p.D123E+p.K166R+p.D177E+p.D192N+p.R211K+p.T215Y+p.V245K+p.K277R+p.R284K+p.T286A+p.E297K |
|||||||||||
| Experimental Note | Identified from the Human Clinical Data | ||||||||||||
| Experiment for Molecule Alteration |
Sanger sequencing assay; GeneSeq assay; Population sequencing of the integrase region assay; DNA sequencing assay; Site-directed mutagenesis | ||||||||||||
| Experiment for Drug Resistance |
PhenoSense assay; Viral load assay; A single-cycle assay; TZM-bl cell line-based phenotypic assay; SDMs and phenotyping assay | ||||||||||||
| Mechanism Description | Some mutations identified were assiacted with resistance in the HIV isolates, those mutations included M41L, A98S, K101P, K102Q, K103N, D123E, K166R, D177E, D192N, R211K, T215Y, V245K, K277R, R284K, T286A, E297K. | ||||||||||||
| Key Molecule: HIV1 Reverse transcriptase (HIV1 RT) | [4] | ||||||||||||
| Resistant Disease | Human immunodeficiency virus infection [ICD-11: 1C62.0] | ||||||||||||
| Molecule Alteration | Missense mutation | p.K20R+p.K64KR+p.L74V+p.K103N+p.V108I+p.Y181C+p.M184V+p.T200A+p.R211K+p.H221Y+p.L228H+p.V245L+p.A272P+p.K277R+p.T286A |
|||||||||||
| Experimental Note | Identified from the Human Clinical Data | ||||||||||||
| Experiment for Molecule Alteration |
Sanger sequencing assay; GeneSeq assay; Population sequencing of the integrase region assay; DNA sequencing assay; Site-directed mutagenesis | ||||||||||||
| Experiment for Drug Resistance |
PhenoSense assay; Viral load assay; A single-cycle assay; TZM-bl cell line-based phenotypic assay; SDMs and phenotyping assay | ||||||||||||
| Mechanism Description | Some mutations identified were assiacted with resistance in the HIV isolates, those mutations included K20R, K64KR, L74V, K103N, V108I, Y181C, M184V, T200A, R211K, H221Y, L228H, V245L, A272P, K277R, T286A. | ||||||||||||
| Key Molecule: HIV1 Reverse transcriptase (HIV1 RT) | [8] | ||||||||||||
| Resistant Disease | Human immunodeficiency virus infection [ICD-11: 1C62.0] | ||||||||||||
| Molecule Alteration | Missense mutation | p.M41L+p.K122E+p.E138G+p.D177E+p.I178L+p.L210W+p.R211K+p.T215Y+p.H221Y+p.F227L+p.M230L+p.V245M+p.I293V |
|||||||||||
| Experimental Note | Identified from the Human Clinical Data | ||||||||||||
| Experiment for Molecule Alteration |
Sanger sequencing assay; GeneSeq assay; Population sequencing of the integrase region assay; DNA sequencing assay; Site-directed mutagenesis | ||||||||||||
| Experiment for Drug Resistance |
PhenoSense assay; Viral load assay; A single-cycle assay; TZM-bl cell line-based phenotypic assay; SDMs and phenotyping assay | ||||||||||||
| Mechanism Description | Some mutations identified were assiacted with resistance in the HIV isolates, those mutations included M41L, K122E, E138G, D177E, I178L, L210W, R211K, T215Y, H221Y, F227L, M230L, V245M, I293V. | ||||||||||||
| Key Molecule: HIV1 Reverse transcriptase (HIV1 RT) | [4] | ||||||||||||
| Resistant Disease | Human immunodeficiency virus infection [ICD-11: 1C62.0] | ||||||||||||
| Molecule Alteration | Missense mutation | p.K20R+p.V35L+p.T39A+p.K101P+p.K103KR+p.K122E+p.I135R+p.D177E+p.I178M+p.T200TA+p.R211K+p.A272P+p.I293V |
|||||||||||
| Experimental Note | Identified from the Human Clinical Data | ||||||||||||
| Experiment for Molecule Alteration |
Sanger sequencing assay; GeneSeq assay; Population sequencing of the integrase region assay; DNA sequencing assay; Site-directed mutagenesis | ||||||||||||
| Experiment for Drug Resistance |
PhenoSense assay; Viral load assay; A single-cycle assay; TZM-bl cell line-based phenotypic assay; SDMs and phenotyping assay | ||||||||||||
| Mechanism Description | Some mutations identified were assiacted with resistance in the HIV isolates, those mutations included K20R, V35L, T39A, K101P, K103KR, K122E, I135R, D177E, I178M, T200TA, R211K, A272P, I293V. | ||||||||||||
| Key Molecule: HIV1 Reverse transcriptase (HIV1 RT) | [8] | ||||||||||||
| Resistant Disease | Human immunodeficiency virus infection [ICD-11: 1C62.0] | ||||||||||||
| Molecule Alteration | Missense mutation | p.V35I+p.M41L+p.D67G+p.L74I+p.L100I+p.K103R+p.K122E+p.V179D+p.M184V+p.I202V+p.R211K+p.T215Y+p.M230L+p.T240K |
|||||||||||
| Experimental Note | Identified from the Human Clinical Data | ||||||||||||
| Experiment for Molecule Alteration |
Sanger sequencing assay; GeneSeq assay; Population sequencing of the integrase region assay; DNA sequencing assay; Site-directed mutagenesis | ||||||||||||
| Experiment for Drug Resistance |
PhenoSense assay; Viral load assay; A single-cycle assay; TZM-bl cell line-based phenotypic assay; SDMs and phenotyping assay | ||||||||||||
| Mechanism Description | Some mutations identified were assiacted with resistance in the HIV isolates, those mutations included V35I, M41L, D67G, L74I, L100I, K103R, K122E, V179D, M184V, I202V, R211K, T215Y, M230L, T240K. | ||||||||||||
| Key Molecule: HIV1 Reverse transcriptase (HIV1 RT) | [4] | ||||||||||||
| Resistant Disease | Human immunodeficiency virus infection [ICD-11: 1C62.0] | ||||||||||||
| Molecule Alteration | Missense mutation | p.K20R+p.V35I+p.K65KR+p.L74LV+p.K103N+p.K122E+p.Y181C+p.T200A+p.R211K+p.F227C+p.M230L+p.V245Q+p.I293V+p.E297A |
|||||||||||
| Experimental Note | Identified from the Human Clinical Data | ||||||||||||
| Experiment for Molecule Alteration |
Sanger sequencing assay; GeneSeq assay; Population sequencing of the integrase region assay; DNA sequencing assay; Site-directed mutagenesis | ||||||||||||
| Experiment for Drug Resistance |
PhenoSense assay; Viral load assay; A single-cycle assay; TZM-bl cell line-based phenotypic assay; SDMs and phenotyping assay | ||||||||||||
| Mechanism Description | Some mutations identified were assiacted with resistance in the HIV isolates, those mutations included K20R, V35I, K65KR, L74LV, K103N, K122E, Y181C, T200A, R211K, F227C, M230L, V245Q, I293V, E297A. | ||||||||||||
| Key Molecule: HIV1 Reverse transcriptase (HIV1 RT) | [4] | ||||||||||||
| Resistant Disease | Human immunodeficiency virus infection [ICD-11: 1C62.0] | ||||||||||||
| Molecule Alteration | Missense mutation | p.M41L+p.K43Q+p.A62AV+p.T69S_ST+p.K101P+p.K103N+p.V118I+p.K122E+p.S162D+p.K166R+p.D177N+p.V179I+p.M184V+p.T200A+p.Q207E+p.L210F+p.R211K+p.T215Y+p.V245E+p.A272P+p.K277R+p.E291D+p.I293V |
|||||||||||
| Experimental Note | Identified from the Human Clinical Data | ||||||||||||
| Experiment for Molecule Alteration |
Sanger sequencing assay; GeneSeq assay; Population sequencing of the integrase region assay; DNA sequencing assay; Site-directed mutagenesis | ||||||||||||
| Experiment for Drug Resistance |
PhenoSense assay; Viral load assay; A single-cycle assay; TZM-bl cell line-based phenotypic assay; SDMs and phenotyping assay | ||||||||||||
| Mechanism Description | Some mutations identified were assiacted with resistance in the HIV isolates, those mutations included M41L, K43Q, A62AV, T69S_ST, K101P, K103N, V118I, K122E, S162D, K166R, D177N, V179I, M184V, T200A, Q207E, L210F, R211K, T215Y, V245E, A272P, K277R, E291D, I293V. | ||||||||||||
| Key Molecule: HIV1 Reverse transcriptase (HIV1 RT) | [4] | ||||||||||||
| Resistant Disease | Human immunodeficiency virus infection [ICD-11: 1C62.0] | ||||||||||||
| Molecule Alteration | Missense mutation | p.T39TP+p.D67N+p.K70R+p.V90I+p.K101P+p.K102R+p.K103N+p.K122E+p.I135V+p.Q151M+p.G196E+p.Q207K+p.R211A+p.K219Q+p.L228LH+p.E248D+p.I293V+p.E297A |
|||||||||||
| Experimental Note | Identified from the Human Clinical Data | ||||||||||||
| Experiment for Molecule Alteration |
Sanger sequencing assay; GeneSeq assay; Population sequencing of the integrase region assay; DNA sequencing assay; Site-directed mutagenesis | ||||||||||||
| Experiment for Drug Resistance |
PhenoSense assay; Viral load assay; A single-cycle assay; TZM-bl cell line-based phenotypic assay; SDMs and phenotyping assay | ||||||||||||
| Mechanism Description | Some mutations identified were assiacted with resistance in the HIV isolates, those mutations included T39TP, D67N, K70R, V90I, K101P, K102R, K103N, K122E, I135V, Q151M, G196E, Q207K, R211A, K219Q, L228LH, E248D, I293V, E297A. | ||||||||||||
| Key Molecule: HIV1 Reverse transcriptase (HIV1 RT) | [9] | ||||||||||||
| Resistant Disease | Human immunodeficiency virus infection [ICD-11: 1C62.0] | ||||||||||||
| Molecule Alteration | Missense mutation | p.L100I+p.K102Q+p.K103R+p.S162C+p.K277R+p.I293V |
|||||||||||
| Experimental Note | Identified from the Human Clinical Data | ||||||||||||
| Experiment for Molecule Alteration |
Sanger sequencing assay; GeneSeq assay; Population sequencing of the integrase region assay; DNA sequencing assay; Site-directed mutagenesis | ||||||||||||
| Experiment for Drug Resistance |
PhenoSense assay; Viral load assay; A single-cycle assay; TZM-bl cell line-based phenotypic assay; SDMs and phenotyping assay | ||||||||||||
| Mechanism Description | Some mutations identified were assiacted with resistance in the HIV isolates, those mutations included L100I, K102Q, K103R, S162C, K277R, I293V. | ||||||||||||
| Key Molecule: HIV1 Reverse transcriptase (HIV1 RT) | [9] | ||||||||||||
| Resistant Disease | Human immunodeficiency virus infection [ICD-11: 1C62.0] | ||||||||||||
| Molecule Alteration | Missense mutation | p.K102Q+p.K103R+p.S162C+p.K277R+p.I293V |
|||||||||||
| Experimental Note | Identified from the Human Clinical Data | ||||||||||||
| Experiment for Molecule Alteration |
Sanger sequencing assay; GeneSeq assay; Population sequencing of the integrase region assay; DNA sequencing assay; Site-directed mutagenesis | ||||||||||||
| Experiment for Drug Resistance |
PhenoSense assay; Viral load assay; A single-cycle assay; TZM-bl cell line-based phenotypic assay; SDMs and phenotyping assay | ||||||||||||
| Mechanism Description | Some mutations identified were assiacted with resistance in the HIV isolates, those mutations included K102Q, K103R, S162C, K277R, I293V. | ||||||||||||
| Key Molecule: HIV1 Reverse transcriptase (HIV1 RT) | [6] | ||||||||||||
| Resistant Disease | Human immunodeficiency virus infection [ICD-11: 1C62.0] | ||||||||||||
| Molecule Alteration | Missense mutation | p.M41L+p.K43N+p.D67G+p.T69N+p.K70R+p.L74I+p.K103N+p.I142V+p.Y181C+p.M184V+p.G196E+p.I202V+p.T215F+p.K219Q+p.V245K+p.K277R+p.A288S+p.I293V |
|||||||||||
| Experimental Note | Identified from the Human Clinical Data | ||||||||||||
| Experiment for Molecule Alteration |
Sanger sequencing assay; GeneSeq assay; Population sequencing of the integrase region assay; DNA sequencing assay; Site-directed mutagenesis | ||||||||||||
| Experiment for Drug Resistance |
PhenoSense assay; Viral load assay; A single-cycle assay; TZM-bl cell line-based phenotypic assay; SDMs and phenotyping assay | ||||||||||||
| Mechanism Description | Some mutations identified were assiacted with resistance in the HIV isolates, those mutations included M41L, K43N, D67G, T69N, K70R, L74I, K103N, I142V, Y181C, M184V, G196E, I202V, T215F, K219Q, V245K, K277R, A288S, I293V. | ||||||||||||
| Key Molecule: HIV1 Reverse transcriptase (HIV1 RT) | [9] | ||||||||||||
| Resistant Disease | Human immunodeficiency virus infection [ICD-11: 1C62.0] | ||||||||||||
| Molecule Alteration | Missense mutation | p.K102Q+p.K103S+p.S162C+p.K277R+p.I293V |
|||||||||||
| Experimental Note | Identified from the Human Clinical Data | ||||||||||||
| Experiment for Molecule Alteration |
Sanger sequencing assay; GeneSeq assay; Population sequencing of the integrase region assay; DNA sequencing assay; Site-directed mutagenesis | ||||||||||||
| Experiment for Drug Resistance |
PhenoSense assay; Viral load assay; A single-cycle assay; TZM-bl cell line-based phenotypic assay; SDMs and phenotyping assay | ||||||||||||
| Mechanism Description | Some mutations identified were assiacted with resistance in the HIV isolates, those mutations included K102Q, K103S, S162C, K277R, I293V. | ||||||||||||
| Key Molecule: HIV1 Reverse transcriptase (HIV1 RT) | [6] | ||||||||||||
| Resistant Disease | Human immunodeficiency virus infection [ICD-11: 1C62.0] | ||||||||||||
| Molecule Alteration | Missense mutation | p.V35R+p.M41L+p.K49R+p.V60I+p.K64H+p.D67N+p.T69N+p.K70R+p.V108I+p.K122Q+p.I135T+p.S162D+p.D177E+p.Y181C+p.M184V+p.V189I+p.T200A+p.T215F+p.D218E+p.K219Q+p.L228R+p.V245E |
|||||||||||
| Experimental Note | Identified from the Human Clinical Data | ||||||||||||
| Experiment for Molecule Alteration |
Sanger sequencing assay; GeneSeq assay; Population sequencing of the integrase region assay; DNA sequencing assay; Site-directed mutagenesis | ||||||||||||
| Experiment for Drug Resistance |
PhenoSense assay; Viral load assay; A single-cycle assay; TZM-bl cell line-based phenotypic assay; SDMs and phenotyping assay | ||||||||||||
| Mechanism Description | Some mutations identified were assiacted with resistance in the HIV isolates, those mutations included V35R, M41L, K49R, V60I, K64H, D67N, T69N, K70R, V108I, K122Q, I135T, S162D, D177E, Y181C, M184V, V189I, T200A, T215F, D218E, K219Q, L228R, V245E. | ||||||||||||
| Key Molecule: HIV1 Reverse transcriptase (HIV1 RT) | [10] | ||||||||||||
| Resistant Disease | Human immunodeficiency virus infection [ICD-11: 1C62.0] | ||||||||||||
| Molecule Alteration | Missense mutation | p.K102Q+p.E138A+p.S162C+p.K277R+p.I293V |
|||||||||||
| Experimental Note | Identified from the Human Clinical Data | ||||||||||||
| Experiment for Molecule Alteration |
Sanger sequencing assay; GeneSeq assay; Population sequencing of the integrase region assay; DNA sequencing assay; Site-directed mutagenesis | ||||||||||||
| Experiment for Drug Resistance |
PhenoSense assay; Viral load assay; A single-cycle assay; TZM-bl cell line-based phenotypic assay; SDMs and phenotyping assay | ||||||||||||
| Mechanism Description | Some mutations identified were assiacted with resistance in the HIV isolates, those mutations included K102Q, E138A, S162C, K277R, I293V. | ||||||||||||
| Key Molecule: HIV1 Reverse transcriptase (HIV1 RT) | [10] | ||||||||||||
| Resistant Disease | Human immunodeficiency virus infection [ICD-11: 1C62.0] | ||||||||||||
| Molecule Alteration | Missense mutation | p.K102Q+p.S162C+p.Y181C+p.K277R+p.I293V |
|||||||||||
| Experimental Note | Identified from the Human Clinical Data | ||||||||||||
| Experiment for Molecule Alteration |
Sanger sequencing assay; GeneSeq assay; Population sequencing of the integrase region assay; DNA sequencing assay; Site-directed mutagenesis | ||||||||||||
| Experiment for Drug Resistance |
PhenoSense assay; Viral load assay; A single-cycle assay; TZM-bl cell line-based phenotypic assay; SDMs and phenotyping assay | ||||||||||||
| Mechanism Description | Some mutations identified were assiacted with resistance in the HIV isolates, those mutations included K102Q, S162C, Y181C, K277R, I293V. | ||||||||||||
| Key Molecule: HIV1 Reverse transcriptase (HIV1 RT) | [10] | ||||||||||||
| Resistant Disease | Human immunodeficiency virus infection [ICD-11: 1C62.0] | ||||||||||||
| Molecule Alteration | Missense mutation | p.K102Q+p.E138G+p.S162C+p.K277R+p.I293V |
|||||||||||
| Experimental Note | Identified from the Human Clinical Data | ||||||||||||
| Experiment for Molecule Alteration |
Sanger sequencing assay; GeneSeq assay; Population sequencing of the integrase region assay; DNA sequencing assay; Site-directed mutagenesis | ||||||||||||
| Experiment for Drug Resistance |
PhenoSense assay; Viral load assay; A single-cycle assay; TZM-bl cell line-based phenotypic assay; SDMs and phenotyping assay | ||||||||||||
| Mechanism Description | Some mutations identified were assiacted with resistance in the HIV isolates, those mutations included K102Q, E138G, S162C, K277R, I293V. | ||||||||||||
| Key Molecule: HIV1 Reverse transcriptase (HIV1 RT) | [6] | ||||||||||||
| Resistant Disease | Human immunodeficiency virus infection [ICD-11: 1C62.0] | ||||||||||||
| Molecule Alteration | Missense mutation | p.K64H+p.D67N+p.T69N+p.K70R+p.V90I+p.K103N+p.V118I+p.I135M+p.T215E+p.K219Q+p.A272P |
|||||||||||
| Experimental Note | Identified from the Human Clinical Data | ||||||||||||
| Experiment for Molecule Alteration |
Sanger sequencing assay; GeneSeq assay; Population sequencing of the integrase region assay; DNA sequencing assay; Site-directed mutagenesis | ||||||||||||
| Experiment for Drug Resistance |
PhenoSense assay; Viral load assay; A single-cycle assay; TZM-bl cell line-based phenotypic assay; SDMs and phenotyping assay | ||||||||||||
| Mechanism Description | Some mutations identified were assiacted with resistance in the HIV isolates, those mutations included K64H, D67N, T69N, K70R, V90I, K103N, V118I, I135M, T215E, K219Q, A272P. | ||||||||||||
| Key Molecule: HIV1 Reverse transcriptase (HIV1 RT) | [10] | ||||||||||||
| Resistant Disease | Human immunodeficiency virus infection [ICD-11: 1C62.0] | ||||||||||||
| Molecule Alteration | Missense mutation | p.K101Q+p.K102Q+p.S162C+p.K277R+p.I293V |
|||||||||||
| Experimental Note | Identified from the Human Clinical Data | ||||||||||||
| Experiment for Molecule Alteration |
Sanger sequencing assay; GeneSeq assay; Population sequencing of the integrase region assay; DNA sequencing assay; Site-directed mutagenesis | ||||||||||||
| Experiment for Drug Resistance |
PhenoSense assay; Viral load assay; A single-cycle assay; TZM-bl cell line-based phenotypic assay; SDMs and phenotyping assay | ||||||||||||
| Mechanism Description | Some mutations identified were assiacted with resistance in the HIV isolates, those mutations included K101Q, K102Q, S162C, K277R, I293V. | ||||||||||||
| Key Molecule: HIV1 Reverse transcriptase (HIV1 RT) | [10] | ||||||||||||
| Resistant Disease | Human immunodeficiency virus infection [ICD-11: 1C62.0] | ||||||||||||
| Molecule Alteration | Missense mutation | p.K102Q+p.E138R+p.S162C+p.K277R+p.I293V |
|||||||||||
| Experimental Note | Identified from the Human Clinical Data | ||||||||||||
| Experiment for Molecule Alteration |
Sanger sequencing assay; GeneSeq assay; Population sequencing of the integrase region assay; DNA sequencing assay; Site-directed mutagenesis | ||||||||||||
| Experiment for Drug Resistance |
PhenoSense assay; Viral load assay; A single-cycle assay; TZM-bl cell line-based phenotypic assay; SDMs and phenotyping assay | ||||||||||||
| Mechanism Description | Some mutations identified were assiacted with resistance in the HIV isolates, those mutations included K102Q, E138R, S162C, K277R, I293V. | ||||||||||||
| Key Molecule: HIV1 Reverse transcriptase (HIV1 RT) | [10] | ||||||||||||
| Resistant Disease | Human immunodeficiency virus infection [ICD-11: 1C62.0] | ||||||||||||
| Molecule Alteration | Missense mutation | p.K101E+p.K102Q+p.S162C+p.M184V+p.K277R+p.I293V |
|||||||||||
| Experimental Note | Identified from the Human Clinical Data | ||||||||||||
| Experiment for Molecule Alteration |
Sanger sequencing assay; GeneSeq assay; Population sequencing of the integrase region assay; DNA sequencing assay; Site-directed mutagenesis | ||||||||||||
| Experiment for Drug Resistance |
PhenoSense assay; Viral load assay; A single-cycle assay; TZM-bl cell line-based phenotypic assay; SDMs and phenotyping assay | ||||||||||||
| Mechanism Description | Some mutations identified were assiacted with resistance in the HIV isolates, those mutations included K101E, K102Q, S162C, M184V, K277R, I293V. | ||||||||||||
| Key Molecule: HIV1 Reverse transcriptase (HIV1 RT) | [6] | ||||||||||||
| Resistant Disease | Human immunodeficiency virus infection [ICD-11: 1C62.0] | ||||||||||||
| Molecule Alteration | Missense mutation | p.V35T+p.K43R+p.K64Y+p.D67N+p.K70R+p.L74I+p.K103S+p.V106A+p.D123E+p.D177E+p.M184V+p.G196E+p.T200A+p.R211S+p.T215F+p.K219Q+p.L228H+p.Q242H+p.K277R+p.R284K |
|||||||||||
| Experimental Note | Identified from the Human Clinical Data | ||||||||||||
| Experiment for Molecule Alteration |
Sanger sequencing assay; GeneSeq assay; Population sequencing of the integrase region assay; DNA sequencing assay; Site-directed mutagenesis | ||||||||||||
| Experiment for Drug Resistance |
PhenoSense assay; Viral load assay; A single-cycle assay; TZM-bl cell line-based phenotypic assay; SDMs and phenotyping assay | ||||||||||||
| Mechanism Description | Some mutations identified were assiacted with resistance in the HIV isolates, those mutations included V35T, K43R, K64Y, D67N, K70R, L74I, K103S, V106A, D123E, D177E, M184V, G196E, T200A, R211S, T215F, K219Q, L228H, Q242H, K277R, R284K. | ||||||||||||
| Key Molecule: HIV1 Reverse transcriptase (HIV1 RT) | [10] | ||||||||||||
| Resistant Disease | Human immunodeficiency virus infection [ICD-11: 1C62.0] | ||||||||||||
| Molecule Alteration | Missense mutation | p.K101E+p.K102Q+p.S162C+p.M184I+p.K277R+p.I293V |
|||||||||||
| Experimental Note | Identified from the Human Clinical Data | ||||||||||||
| Experiment for Molecule Alteration |
Sanger sequencing assay; GeneSeq assay; Population sequencing of the integrase region assay; DNA sequencing assay; Site-directed mutagenesis | ||||||||||||
| Experiment for Drug Resistance |
PhenoSense assay; Viral load assay; A single-cycle assay; TZM-bl cell line-based phenotypic assay; SDMs and phenotyping assay | ||||||||||||
| Mechanism Description | Some mutations identified were assiacted with resistance in the HIV isolates, those mutations included K101E, K102Q, S162C, M184I, K277R, I293V. | ||||||||||||
| Key Molecule: HIV1 Reverse transcriptase (HIV1 RT) | [10] | ||||||||||||
| Resistant Disease | Human immunodeficiency virus infection [ICD-11: 1C62.0] | ||||||||||||
| Molecule Alteration | Missense mutation | p.K101E+p.K102Q+p.S162C+p.K277R+p.I293V |
|||||||||||
| Experimental Note | Identified from the Human Clinical Data | ||||||||||||
| Experiment for Molecule Alteration |
Sanger sequencing assay; GeneSeq assay; Population sequencing of the integrase region assay; DNA sequencing assay; Site-directed mutagenesis | ||||||||||||
| Experiment for Drug Resistance |
PhenoSense assay; Viral load assay; A single-cycle assay; TZM-bl cell line-based phenotypic assay; SDMs and phenotyping assay | ||||||||||||
| Mechanism Description | Some mutations identified were assiacted with resistance in the HIV isolates, those mutations included K101E, K102Q, S162C, K277R, I293V. | ||||||||||||
| Key Molecule: HIV1 Reverse transcriptase (HIV1 RT) | [6] | ||||||||||||
| Resistant Disease | Human immunodeficiency virus infection [ICD-11: 1C62.0] | ||||||||||||
| Molecule Alteration | Missense mutation | p.V35R+p.M41L+p.K49R+p.V60I+p.D67N+p.T69N+p.K70R+p.V108I+p.K122Q+p.I135T+p.S162D+p.D177E+p.Y181C+p.M184V+p.V189I+p.T200A+p.T215F+p.D218E+p.K219Q+p.L228R+p.V245E+p.I293V |
|||||||||||
| Experimental Note | Identified from the Human Clinical Data | ||||||||||||
| Experiment for Molecule Alteration |
Sanger sequencing assay; GeneSeq assay; Population sequencing of the integrase region assay; DNA sequencing assay; Site-directed mutagenesis | ||||||||||||
| Experiment for Drug Resistance |
PhenoSense assay; Viral load assay; A single-cycle assay; TZM-bl cell line-based phenotypic assay; SDMs and phenotyping assay | ||||||||||||
| Mechanism Description | Some mutations identified were assiacted with resistance in the HIV isolates, those mutations included V35R, M41L, K49R, V60I, D67N, T69N, K70R, V108I, K122Q, I135T, S162D, D177E, Y181C, M184V, V189I, T200A, T215F, D218E, K219Q, L228R, V245E, I293V. | ||||||||||||
| Key Molecule: HIV1 Reverse transcriptase (HIV1 RT) | [9] | ||||||||||||
| Resistant Disease | Human immunodeficiency virus infection [ICD-11: 1C62.0] | ||||||||||||
| Molecule Alteration | Missense mutation | p.K102Q+p.K103R+p.S162C+p.V179D+p.K277R+p.I293V |
|||||||||||
| Experimental Note | Identified from the Human Clinical Data | ||||||||||||
| Experiment for Molecule Alteration |
Sanger sequencing assay; GeneSeq assay; Population sequencing of the integrase region assay; DNA sequencing assay; Site-directed mutagenesis | ||||||||||||
| Experiment for Drug Resistance |
PhenoSense assay; Viral load assay; A single-cycle assay; TZM-bl cell line-based phenotypic assay; SDMs and phenotyping assay | ||||||||||||
| Mechanism Description | Some mutations identified were assiacted with resistance in the HIV isolates, those mutations included K102Q, K103R, S162C, V179D, K277R, I293V. | ||||||||||||
| Key Molecule: HIV1 Reverse transcriptase (HIV1 RT) | [10] | ||||||||||||
| Resistant Disease | Human immunodeficiency virus infection [ICD-11: 1C62.0] | ||||||||||||
| Molecule Alteration | Missense mutation | p.K102Q+p.E138Q+p.S162C+p.K277R+p.I293V |
|||||||||||
| Experimental Note | Identified from the Human Clinical Data | ||||||||||||
| Experiment for Molecule Alteration |
Sanger sequencing assay; GeneSeq assay; Population sequencing of the integrase region assay; DNA sequencing assay; Site-directed mutagenesis | ||||||||||||
| Experiment for Drug Resistance |
PhenoSense assay; Viral load assay; A single-cycle assay; TZM-bl cell line-based phenotypic assay; SDMs and phenotyping assay | ||||||||||||
| Mechanism Description | Some mutations identified were assiacted with resistance in the HIV isolates, those mutations included K102Q, E138Q, S162C, K277R, I293V. | ||||||||||||
| Key Molecule: HIV1 Reverse transcriptase (HIV1 RT) | [11] | ||||||||||||
| Resistant Disease | Human immunodeficiency virus infection [ICD-11: 1C62.0] | ||||||||||||
| Molecule Alteration | Missense mutation | p.Y188L |
|||||||||||
| Experimental Note | Identified from the Human Clinical Data | ||||||||||||
| Experiment for Molecule Alteration |
Sanger sequencing assay; GeneSeq assay; Population sequencing of the integrase region assay; DNA sequencing assay; Site-directed mutagenesis | ||||||||||||
| Experiment for Drug Resistance |
PhenoSense assay; Viral load assay; A single-cycle assay; TZM-bl cell line-based phenotypic assay; SDMs and phenotyping assay | ||||||||||||
| Mechanism Description | Some mutations identified were assiacted with resistance in the HIV isolates, those mutations included Y188L. | ||||||||||||
| Key Molecule: HIV1 Reverse transcriptase (HIV1 RT) | [9] | ||||||||||||
| Resistant Disease | Human immunodeficiency virus infection [ICD-11: 1C62.0] | ||||||||||||
| Molecule Alteration | Missense mutation | p.L100I+p.K102Q+p.K103R+p.S162C+p.V179D+p.K277R+p.I293V |
|||||||||||
| Experimental Note | Identified from the Human Clinical Data | ||||||||||||
| Experiment for Molecule Alteration |
Sanger sequencing assay; GeneSeq assay; Population sequencing of the integrase region assay; DNA sequencing assay; Site-directed mutagenesis | ||||||||||||
| Experiment for Drug Resistance |
PhenoSense assay; Viral load assay; A single-cycle assay; TZM-bl cell line-based phenotypic assay; SDMs and phenotyping assay | ||||||||||||
| Mechanism Description | Some mutations identified were assiacted with resistance in the HIV isolates, those mutations included L100I, K102Q, K103R, S162C, V179D, K277R, I293V. | ||||||||||||
| Key Molecule: HIV1 Reverse transcriptase (HIV1 RT) | [10] | ||||||||||||
| Resistant Disease | Human immunodeficiency virus infection [ICD-11: 1C62.0] | ||||||||||||
| Molecule Alteration | Missense mutation | p.K102Q+p.S162C+p.Y181I+p.K277R+p.I293V |
|||||||||||
| Experimental Note | Identified from the Human Clinical Data | ||||||||||||
| Experiment for Molecule Alteration |
Sanger sequencing assay; GeneSeq assay; Population sequencing of the integrase region assay; DNA sequencing assay; Site-directed mutagenesis | ||||||||||||
| Experiment for Drug Resistance |
PhenoSense assay; Viral load assay; A single-cycle assay; TZM-bl cell line-based phenotypic assay; SDMs and phenotyping assay | ||||||||||||
| Mechanism Description | Some mutations identified were assiacted with resistance in the HIV isolates, those mutations included K102Q, S162C, Y181I, K277R, I293V. | ||||||||||||
| Key Molecule: HIV1 Reverse transcriptase (HIV1 RT) | [10] | ||||||||||||
| Resistant Disease | Human immunodeficiency virus infection [ICD-11: 1C62.0] | ||||||||||||
| Molecule Alteration | Missense mutation | p.K101P+p.K102Q+p.S162C+p.K277R+p.I293V |
|||||||||||
| Experimental Note | Identified from the Human Clinical Data | ||||||||||||
| Experiment for Molecule Alteration |
Sanger sequencing assay; GeneSeq assay; Population sequencing of the integrase region assay; DNA sequencing assay; Site-directed mutagenesis | ||||||||||||
| Experiment for Drug Resistance |
PhenoSense assay; Viral load assay; A single-cycle assay; TZM-bl cell line-based phenotypic assay; SDMs and phenotyping assay | ||||||||||||
| Mechanism Description | Some mutations identified were assiacted with resistance in the HIV isolates, those mutations included K101P, K102Q, S162C, K277R, I293V. | ||||||||||||
| Key Molecule: HIV1 Reverse transcriptase (HIV1 RT) | [9] | ||||||||||||
| Resistant Disease | Human immunodeficiency virus infection [ICD-11: 1C62.0] | ||||||||||||
| Molecule Alteration | Missense mutation | p.L100I+p.K102Q+p.K103N+p.S162C+p.K277R+p.I293V |
|||||||||||
| Experimental Note | Identified from the Human Clinical Data | ||||||||||||
| Experiment for Molecule Alteration |
Sanger sequencing assay; GeneSeq assay; Population sequencing of the integrase region assay; DNA sequencing assay; Site-directed mutagenesis | ||||||||||||
| Experiment for Drug Resistance |
PhenoSense assay; Viral load assay; A single-cycle assay; TZM-bl cell line-based phenotypic assay; SDMs and phenotyping assay | ||||||||||||
| Mechanism Description | Some mutations identified were assiacted with resistance in the HIV isolates, those mutations included L100I, K102Q, K103N, S162C, K277R, I293V. | ||||||||||||
| Key Molecule: HIV1 Reverse transcriptase (HIV1 RT) | [9] | ||||||||||||
| Resistant Disease | Human immunodeficiency virus infection [ICD-11: 1C62.0] | ||||||||||||
| Molecule Alteration | Missense mutation | p.L100I+p.K102Q+p.K103S+p.S162C+p.K277R+p.I293V |
|||||||||||
| Experimental Note | Identified from the Human Clinical Data | ||||||||||||
| Experiment for Molecule Alteration |
Sanger sequencing assay; GeneSeq assay; Population sequencing of the integrase region assay; DNA sequencing assay; Site-directed mutagenesis | ||||||||||||
| Experiment for Drug Resistance |
PhenoSense assay; Viral load assay; A single-cycle assay; TZM-bl cell line-based phenotypic assay; SDMs and phenotyping assay | ||||||||||||
| Mechanism Description | Some mutations identified were assiacted with resistance in the HIV isolates, those mutations included L100I, K102Q, K103S, S162C, K277R, I293V. | ||||||||||||
| Drug Sensitivity Data Categorized by Their Corresponding Mechanisms | |||||||||||||
|
|
|||||||||||||
| Key Molecule: HIV1 Reverse transcriptase (HIV1 RT) | [3] | ||||||||||||
| Sensitive Disease | Human immunodeficiency virus infection [ICD-11: 1C62.0] | ||||||||||||
| Molecule Alteration | Missense mutation | p.S48T+p.R211N+p.V245N+p.L283I+p.V292I+p.I293V |
|||||||||||
| Wild Type Structure | Method: X-ray diffraction | Resolution: 2.09 Å | |||||||||||
| Mutant Type Structure | Method: AlphaFold | Average pLDDT: 87.034 |

|
||||||||||
| Download The Information of Sequence | Download The Structure File | ||||||||||||
-
P
P
I
I
590
|
S
S
P
P
I
I
E
E
T
T
V
V
P
P
V
V
K
K
L
L
600
|
K
K
P
P
G
G
M
M
D
D
G
G
P
P
K
K
V
V
K
K
610
|
Q
Q
W
W
P
P
L
L
T
T
E
E
E
E
K
K
I
I
K
K
620
|
A
A
L
L
V
V
E
E
I
I
C
C
T
T
E
E
M
M
E
E
630
|
K
K
E
E
G
G
K
K
I
I
S
T
K
K
I
I
G
G
P
P
640
|
E
E
N
N
P
P
Y
Y
N
N
T
T
P
P
V
V
F
F
A
A
650
|
I
I
K
K
K
K
K
K
D
D
S
S
T
T
K
K
W
W
R
R
660
|
K
K
L
L
V
V
D
D
F
F
R
R
E
E
L
L
N
N
K
K
670
|
R
R
T
T
Q
Q
D
D
F
F
W
W
E
E
V
V
Q
Q
L
L
680
|
G
G
I
I
P
P
H
H
P
P
A
A
G
G
L
L
K
K
K
K
690
|
K
K
K
K
S
S
V
V
T
T
V
V
L
L
D
D
V
V
G
G
700
|
D
D
A
A
Y
Y
F
F
S
S
V
V
P
P
L
L
D
D
E
E
710
|
D
D
F
F
R
R
K
K
Y
Y
T
T
A
A
F
F
T
T
I
I
720
|
P
P
S
S
I
I
N
N
N
N
E
E
T
T
P
P
G
G
I
I
730
|
R
R
Y
Y
Q
Q
Y
Y
N
N
V
V
L
L
P
P
Q
Q
G
G
740
|
W
W
K
K
G
G
S
S
P
P
A
A
I
I
F
F
Q
Q
S
S
750
|
S
S
M
M
T
T
K
K
I
I
L
L
E
E
P
P
F
F
R
R
760
|
K
K
Q
Q
N
N
P
P
D
D
I
I
V
V
I
I
Y
Y
Q
Q
770
|
Y
Y
M
M
D
D
D
D
L
L
Y
Y
V
V
G
G
S
S
D
D
780
|
L
L
E
E
I
I
G
G
Q
Q
H
H
R
R
T
T
K
K
I
I
790
|
E
E
E
E
L
L
R
R
Q
Q
H
H
L
L
L
L
R
N
W
W
800
|
G
G
L
L
T
T
T
T
P
P
D
D
K
K
K
K
H
H
Q
Q
810
|
K
K
E
E
P
P
P
P
F
F
L
L
W
W
M
M
G
G
Y
Y
820
|
E
E
L
L
H
H
P
P
D
D
K
K
W
W
T
T
V
V
Q
Q
830
|
P
P
I
I
V
N
L
L
P
P
E
E
K
K
D
D
S
S
W
W
840
|
T
T
V
V
N
N
D
D
I
I
Q
Q
K
K
L
L
V
V
G
G
850
|
K
K
L
L
N
N
W
W
A
A
S
S
Q
Q
I
I
Y
Y
P
P
860
|
G
G
I
I
K
K
V
V
R
R
Q
Q
L
L
C
C
K
K
L
L
870
|
L
I
R
R
G
G
T
T
K
K
A
A
L
L
T
T
E
E
V
I
880
|
I
V
P
P
L
L
T
T
E
E
E
E
A
A
E
E
L
L
E
E
890
|
L
L
A
A
E
E
N
N
R
R
E
E
I
I
L
L
K
K
E
E
900
|
P
P
V
V
H
H
G
G
V
V
Y
Y
Y
Y
D
D
P
P
S
S
910
|
K
K
D
D
L
L
I
I
A
A
E
E
I
I
Q
Q
K
K
Q
Q
920
|
G
G
Q
Q
G
G
Q
Q
W
W
T
T
Y
Y
Q
Q
I
I
Y
Y
930
|
Q
Q
E
E
P
P
F
F
K
K
N
N
L
L
K
K
T
T
G
G
940
|
K
K
Y
Y
A
A
R
R
M
M
R
R
G
G
A
A
H
H
T
T
950
|
N
N
D
D
V
V
K
K
Q
Q
L
L
T
T
E
E
A
A
V
V
960
|
Q
Q
K
K
I
I
T
T
T
T
E
E
S
S
I
I
V
V
I
I
970
|
W
W
G
G
K
K
T
T
P
P
K
K
F
F
K
K
L
L
P
P
980
|
I
I
Q
Q
K
K
E
E
T
T
W
W
E
E
T
T
W
W
W
W
990
|
T
T
E
E
Y
Y
W
W
Q
Q
A
A
T
T
W
W
I
I
P
P
1000
|
E
E
W
W
E
E
F
F
V
V
N
N
T
T
P
P
P
P
L
L
1010
|
V
V
K
K
L
L
W
W
Y
Y
Q
Q
L
L
E
E
K
K
E
E
1020
|
P
P
I
I
V
V
G
G
A
A
E
E
T
T
F
F
Y
Y
V
V
1030
|
D
D
G
G
A
A
A
A
N
N
R
R
E
E
T
T
K
K
L
L
1040
|
G
G
K
K
A
A
G
G
Y
Y
V
V
T
T
N
N
R
R
G
G
1050
|
R
R
Q
Q
K
K
V
V
V
V
T
T
L
L
T
T
D
D
T
T
1060
|
T
T
N
N
Q
Q
K
K
T
T
E
E
L
L
Q
Q
A
A
I
I
1070
|
Y
Y
L
L
A
A
L
L
Q
Q
D
D
S
S
G
G
L
L
E
E
1080
|
V
V
N
N
I
I
V
V
T
T
D
D
S
S
Q
Q
Y
Y
A
A
1090
|
L
L
G
G
I
I
I
I
Q
Q
A
A
Q
Q
P
P
D
D
Q
Q
1100
|
S
S
E
E
S
S
E
E
L
L
V
V
N
N
Q
Q
I
I
I
I
1110
|
E
E
Q
Q
L
L
I
I
K
K
K
K
E
E
K
K
V
V
Y
Y
1120
|
L
L
A
A
W
W
V
V
P
P
A
A
H
H
K
K
G
G
I
I
1130
|
G
G
G
G
N
N
E
E
Q
Q
V
V
D
D
K
K
L
L
V
V
1140
|
S
S
A
A
G
G
I
I
R
R
K
K
V
V
L
L
|
|||||||||||||
| Experimental Note | Identified from the Human Clinical Data | ||||||||||||
| Experiment for Molecule Alteration |
GeneSeq assay; PhenoSense GT assay | ||||||||||||
| Experiment for Drug Resistance |
Viral load assay | ||||||||||||
| Mechanism Description | Amino acid changes in IN may contribute to drug resistance or sensitivity. | ||||||||||||
| Key Molecule: HIV1 Reverse transcriptase (HIV1 RT) | [3] | ||||||||||||
| Sensitive Disease | Human immunodeficiency virus infection [ICD-11: 1C62.0] | ||||||||||||
| Molecule Alteration | Missense mutation | p.K11KR+p.V35I+p.R83K+p.T200TA+p.Q207A+p.R211K+p.I293V |
|||||||||||
| Wild Type Structure | Method: X-ray diffraction | Resolution: 2.09 Å | |||||||||||
| Mutant Type Structure | Method: AlphaFold | Average pLDDT: 87.008 |

|
||||||||||
| Download The Information of Sequence | Download The Structure File | ||||||||||||
-
P
P
I
I
590
|
S
S
P
P
I
I
E
E
T
T
V
V
P
P
V
V
K
R
L
L
600
|
K
K
P
P
G
G
M
M
D
D
G
G
P
P
K
K
V
V
K
K
610
|
Q
Q
W
W
P
P
L
L
T
T
E
E
E
E
K
K
I
I
K
K
620
|
A
A
L
L
V
I
E
E
I
I
C
C
T
T
E
E
M
M
E
E
630
|
K
K
E
E
G
G
K
K
I
I
S
S
K
K
I
I
G
G
P
P
640
|
E
E
N
N
P
P
Y
Y
N
N
T
T
P
P
V
V
F
F
A
A
650
|
I
I
K
K
K
K
K
K
D
D
S
S
T
T
K
K
W
W
R
R
660
|
K
K
L
L
V
V
D
D
F
F
R
R
E
E
L
L
N
N
K
K
670
|
R
K
T
T
Q
Q
D
D
F
F
W
W
E
E
V
V
Q
Q
L
L
680
|
G
G
I
I
P
P
H
H
P
P
A
A
G
G
L
L
K
K
K
K
690
|
K
K
K
K
S
S
V
V
T
T
V
V
L
L
D
D
V
V
G
G
700
|
D
D
A
A
Y
Y
F
F
S
S
V
V
P
P
L
L
D
D
E
E
710
|
D
D
F
F
R
R
K
K
Y
Y
T
T
A
A
F
F
T
T
I
I
720
|
P
P
S
S
I
I
N
N
N
N
E
E
T
T
P
P
G
G
I
I
730
|
R
R
Y
Y
Q
Q
Y
Y
N
N
V
V
L
L
P
P
Q
Q
G
G
740
|
W
W
K
K
G
G
S
S
P
P
A
A
I
I
F
F
Q
Q
S
S
750
|
S
S
M
M
T
T
K
K
I
I
L
L
E
E
P
P
F
F
R
R
760
|
K
K
Q
Q
N
N
P
P
D
D
I
I
V
V
I
I
Y
Y
Q
Q
770
|
Y
Y
M
M
D
D
D
D
L
L
Y
Y
V
V
G
G
S
S
D
D
780
|
L
L
E
E
I
I
G
G
Q
Q
H
H
R
R
T
A
K
K
I
I
790
|
E
E
E
E
L
L
R
R
Q
A
H
H
L
L
L
L
R
K
W
W
800
|
G
G
L
L
T
T
T
T
P
P
D
D
K
K
K
K
H
H
Q
Q
810
|
K
K
E
E
P
P
P
P
F
F
L
L
W
W
M
M
G
G
Y
Y
820
|
E
E
L
L
H
H
P
P
D
D
K
K
W
W
T
T
V
V
Q
Q
830
|
P
P
I
I
V
V
L
L
P
P
E
E
K
K
D
D
S
S
W
W
840
|
T
T
V
V
N
N
D
D
I
I
Q
Q
K
K
L
L
V
V
G
G
850
|
K
K
L
L
N
N
W
W
A
A
S
S
Q
Q
I
I
Y
Y
P
P
860
|
G
G
I
I
K
K
V
V
R
R
Q
Q
L
L
C
C
K
K
L
L
870
|
L
L
R
R
G
G
T
T
K
K
A
A
L
L
T
T
E
E
V
V
880
|
I
V
P
P
L
L
T
T
E
E
E
E
A
A
E
E
L
L
E
E
890
|
L
L
A
A
E
E
N
N
R
R
E
E
I
I
L
L
K
K
E
E
900
|
P
P
V
V
H
H
G
G
V
V
Y
Y
Y
Y
D
D
P
P
S
S
910
|
K
K
D
D
L
L
I
I
A
A
E
E
I
I
Q
Q
K
K
Q
Q
920
|
G
G
Q
Q
G
G
Q
Q
W
W
T
T
Y
Y
Q
Q
I
I
Y
Y
930
|
Q
Q
E
E
P
P
F
F
K
K
N
N
L
L
K
K
T
T
G
G
940
|
K
K
Y
Y
A
A
R
R
M
M
R
R
G
G
A
A
H
H
T
T
950
|
N
N
D
D
V
V
K
K
Q
Q
L
L
T
T
E
E
A
A
V
V
960
|
Q
Q
K
K
I
I
T
T
T
T
E
E
S
S
I
I
V
V
I
I
970
|
W
W
G
G
K
K
T
T
P
P
K
K
F
F
K
K
L
L
P
P
980
|
I
I
Q
Q
K
K
E
E
T
T
W
W
E
E
T
T
W
W
W
W
990
|
T
T
E
E
Y
Y
W
W
Q
Q
A
A
T
T
W
W
I
I
P
P
1000
|
E
E
W
W
E
E
F
F
V
V
N
N
T
T
P
P
P
P
L
L
1010
|
V
V
K
K
L
L
W
W
Y
Y
Q
Q
L
L
E
E
K
K
E
E
1020
|
P
P
I
I
V
V
G
G
A
A
E
E
T
T
F
F
Y
Y
V
V
1030
|
D
D
G
G
A
A
A
A
N
N
R
R
E
E
T
T
K
K
L
L
1040
|
G
G
K
K
A
A
G
G
Y
Y
V
V
T
T
N
N
R
R
G
G
1050
|
R
R
Q
Q
K
K
V
V
V
V
T
T
L
L
T
T
D
D
T
T
1060
|
T
T
N
N
Q
Q
K
K
T
T
E
E
L
L
Q
Q
A
A
I
I
1070
|
Y
Y
L
L
A
A
L
L
Q
Q
D
D
S
S
G
G
L
L
E
E
1080
|
V
V
N
N
I
I
V
V
T
T
D
D
S
S
Q
Q
Y
Y
A
A
1090
|
L
L
G
G
I
I
I
I
Q
Q
A
A
Q
Q
P
P
D
D
Q
Q
1100
|
S
S
E
E
S
S
E
E
L
L
V
V
N
N
Q
Q
I
I
I
I
1110
|
E
E
Q
Q
L
L
I
I
K
K
K
K
E
E
K
K
V
V
Y
Y
1120
|
L
L
A
A
W
W
V
V
P
P
A
A
H
H
K
K
G
G
I
I
1130
|
G
G
G
G
N
N
E
E
Q
Q
V
V
D
D
K
K
L
L
V
V
1140
|
S
S
A
A
G
G
I
I
R
R
K
K
V
V
L
L
|
|||||||||||||
| Experimental Note | Identified from the Human Clinical Data | ||||||||||||
| Experiment for Molecule Alteration |
GeneSeq assay; PhenoSense GT assay | ||||||||||||
| Experiment for Drug Resistance |
Viral load assay | ||||||||||||
| Mechanism Description | Amino acid changes in IN may contribute to drug resistance or sensitivity. | ||||||||||||
| Key Molecule: HIV1 Reverse transcriptase (HIV1 RT) | [3] | ||||||||||||
| Sensitive Disease | Human immunodeficiency virus infection [ICD-11: 1C62.0] | ||||||||||||
| Molecule Alteration | Missense mutation | p.V35I+p.R83K+p.D177E+p.G196E+p.H208HY+p.L210LF+p.R211K+p.E291D+p.E297K |
|||||||||||
| Wild Type Structure | Method: X-ray diffraction | Resolution: 2.09 Å | |||||||||||
| Mutant Type Structure | Method: AlphaFold | Average pLDDT: 86.859 |

|
||||||||||
| Download The Information of Sequence | Download The Structure File | ||||||||||||
-
P
P
I
I
590
|
S
S
P
P
I
I
E
E
T
T
V
V
P
P
V
V
K
K
L
L
600
|
K
K
P
P
G
G
M
M
D
D
G
G
P
P
K
K
V
V
K
K
610
|
Q
Q
W
W
P
P
L
L
T
T
E
E
E
E
K
K
I
I
K
K
620
|
A
A
L
L
V
I
E
E
I
I
C
C
T
T
E
E
M
M
E
E
630
|
K
K
E
E
G
G
K
K
I
I
S
S
K
K
I
I
G
G
P
P
640
|
E
E
N
N
P
P
Y
Y
N
N
T
T
P
P
V
V
F
F
A
A
650
|
I
I
K
K
K
K
K
K
D
D
S
S
T
T
K
K
W
W
R
R
660
|
K
K
L
L
V
V
D
D
F
F
R
R
E
E
L
L
N
N
K
K
670
|
R
K
T
T
Q
Q
D
D
F
F
W
W
E
E
V
V
Q
Q
L
L
680
|
G
G
I
I
P
P
H
H
P
P
A
A
G
G
L
L
K
K
K
K
690
|
K
K
K
K
S
S
V
V
T
T
V
V
L
L
D
D
V
V
G
G
700
|
D
D
A
A
Y
Y
F
F
S
S
V
V
P
P
L
L
D
D
E
E
710
|
D
D
F
F
R
R
K
K
Y
Y
T
T
A
A
F
F
T
T
I
I
720
|
P
P
S
S
I
I
N
N
N
N
E
E
T
T
P
P
G
G
I
I
730
|
R
R
Y
Y
Q
Q
Y
Y
N
N
V
V
L
L
P
P
Q
Q
G
G
740
|
W
W
K
K
G
G
S
S
P
P
A
A
I
I
F
F
Q
Q
S
S
750
|
S
S
M
M
T
T
K
K
I
I
L
L
E
E
P
P
F
F
R
R
760
|
K
K
Q
Q
N
N
P
P
D
E
I
I
V
V
I
I
Y
Y
Q
Q
770
|
Y
Y
M
M
D
D
D
D
L
L
Y
Y
V
V
G
G
S
S
D
D
780
|
L
L
E
E
I
I
G
E
Q
Q
H
H
R
R
T
T
K
K
I
I
790
|
E
E
E
E
L
L
R
R
Q
Q
H
Y
L
L
L
F
R
K
W
W
800
|
G
G
L
L
T
T
T
T
P
P
D
D
K
K
K
K
H
H
Q
Q
810
|
K
K
E
E
P
P
P
P
F
F
L
L
W
W
M
M
G
G
Y
Y
820
|
E
E
L
L
H
H
P
P
D
D
K
K
W
W
T
T
V
V
Q
Q
830
|
P
P
I
I
V
V
L
L
P
P
E
E
K
K
D
D
S
S
W
W
840
|
T
T
V
V
N
N
D
D
I
I
Q
Q
K
K
L
L
V
V
G
G
850
|
K
K
L
L
N
N
W
W
A
A
S
S
Q
Q
I
I
Y
Y
P
P
860
|
G
G
I
I
K
K
V
V
R
R
Q
Q
L
L
C
C
K
K
L
L
870
|
L
L
R
R
G
G
T
T
K
K
A
A
L
L
T
T
E
D
V
V
880
|
I
I
P
P
L
L
T
T
E
K
E
E
A
A
E
E
L
L
E
E
890
|
L
L
A
A
E
E
N
N
R
R
E
E
I
I
L
L
K
K
E
E
900
|
P
P
V
V
H
H
G
G
V
V
Y
Y
Y
Y
D
D
P
P
S
S
910
|
K
K
D
D
L
L
I
I
A
A
E
E
I
I
Q
Q
K
K
Q
Q
920
|
G
G
Q
Q
G
G
Q
Q
W
W
T
T
Y
Y
Q
Q
I
I
Y
Y
930
|
Q
Q
E
E
P
P
F
F
K
K
N
N
L
L
K
K
T
T
G
G
940
|
K
K
Y
Y
A
A
R
R
M
M
R
R
G
G
A
A
H
H
T
T
950
|
N
N
D
D
V
V
K
K
Q
Q
L
L
T
T
E
E
A
A
V
V
960
|
Q
Q
K
K
I
I
T
T
T
T
E
E
S
S
I
I
V
V
I
I
970
|
W
W
G
G
K
K
T
T
P
P
K
K
F
F
K
K
L
L
P
P
980
|
I
I
Q
Q
K
K
E
E
T
T
W
W
E
E
T
T
W
W
W
W
990
|
T
T
E
E
Y
Y
W
W
Q
Q
A
A
T
T
W
W
I
I
P
P
1000
|
E
E
W
W
E
E
F
F
V
V
N
N
T
T
P
P
P
P
L
L
1010
|
V
V
K
K
L
L
W
W
Y
Y
Q
Q
L
L
E
E
K
K
E
E
1020
|
P
P
I
I
V
V
G
G
A
A
E
E
T
T
F
F
Y
Y
V
V
1030
|
D
D
G
G
A
A
A
A
N
N
R
R
E
E
T
T
K
K
L
L
1040
|
G
G
K
K
A
A
G
G
Y
Y
V
V
T
T
N
N
R
R
G
G
1050
|
R
R
Q
Q
K
K
V
V
V
V
T
T
L
L
T
T
D
D
T
T
1060
|
T
T
N
N
Q
Q
K
K
T
T
E
E
L
L
Q
Q
A
A
I
I
1070
|
Y
Y
L
L
A
A
L
L
Q
Q
D
D
S
S
G
G
L
L
E
E
1080
|
V
V
N
N
I
I
V
V
T
T
D
D
S
S
Q
Q
Y
Y
A
A
1090
|
L
L
G
G
I
I
I
I
Q
Q
A
A
Q
Q
P
P
D
D
Q
Q
1100
|
S
S
E
E
S
S
E
E
L
L
V
V
N
N
Q
Q
I
I
I
I
1110
|
E
E
Q
Q
L
L
I
I
K
K
K
K
E
E
K
K
V
V
Y
Y
1120
|
L
L
A
A
W
W
V
V
P
P
A
A
H
H
K
K
G
G
I
I
1130
|
G
G
G
G
N
N
E
E
Q
Q
V
V
D
D
K
K
L
L
V
V
1140
|
S
S
A
A
G
G
I
I
R
R
K
K
V
V
L
L
|
|||||||||||||
| Experimental Note | Identified from the Human Clinical Data | ||||||||||||
| Experiment for Molecule Alteration |
GeneSeq assay; PhenoSense GT assay | ||||||||||||
| Experiment for Drug Resistance |
Viral load assay | ||||||||||||
| Mechanism Description | Amino acid changes in IN may contribute to drug resistance or sensitivity. | ||||||||||||
| Key Molecule: HIV1 Reverse transcriptase (HIV1 RT) | [2] | ||||||||||||
| Sensitive Disease | Human immunodeficiency virus infection [ICD-11: 1C62.0] | ||||||||||||
| Molecule Alteration | Missense mutation | p.V35I+p.R83K+p.S162A+p.I178IM+p.Q207E+p.I293V+p.E297A+p.L301M |
|||||||||||
| Wild Type Structure | Method: X-ray diffraction | Resolution: 2.09 Å | |||||||||||
| Mutant Type Structure | Method: AlphaFold | Average pLDDT: 86.996 |

|
||||||||||
| Download The Information of Sequence | Download The Structure File | ||||||||||||
-
P
P
I
I
590
|
S
S
P
P
I
I
E
E
T
T
V
V
P
P
V
V
K
K
L
L
600
|
K
K
P
P
G
G
M
M
D
D
G
G
P
P
K
K
V
V
K
K
610
|
Q
Q
W
W
P
P
L
L
T
T
E
E
E
E
K
K
I
I
K
K
620
|
A
A
L
L
V
I
E
E
I
I
C
C
T
T
E
E
M
M
E
E
630
|
K
K
E
E
G
G
K
K
I
I
S
S
K
K
I
I
G
G
P
P
640
|
E
E
N
N
P
P
Y
Y
N
N
T
T
P
P
V
V
F
F
A
A
650
|
I
I
K
K
K
K
K
K
D
D
S
S
T
T
K
K
W
W
R
R
660
|
K
K
L
L
V
V
D
D
F
F
R
R
E
E
L
L
N
N
K
K
670
|
R
K
T
T
Q
Q
D
D
F
F
W
W
E
E
V
V
Q
Q
L
L
680
|
G
G
I
I
P
P
H
H
P
P
A
A
G
G
L
L
K
K
K
K
690
|
K
K
K
K
S
S
V
V
T
T
V
V
L
L
D
D
V
V
G
G
700
|
D
D
A
A
Y
Y
F
F
S
S
V
V
P
P
L
L
D
D
E
E
710
|
D
D
F
F
R
R
K
K
Y
Y
T
T
A
A
F
F
T
T
I
I
720
|
P
P
S
S
I
I
N
N
N
N
E
E
T
T
P
P
G
G
I
I
730
|
R
R
Y
Y
Q
Q
Y
Y
N
N
V
V
L
L
P
P
Q
Q
G
G
740
|
W
W
K
K
G
G
S
S
P
P
A
A
I
I
F
F
Q
Q
S
A
750
|
S
S
M
M
T
T
K
K
I
I
L
L
E
E
P
P
F
F
R
R
760
|
K
K
Q
Q
N
N
P
P
D
D
I
M
V
V
I
I
Y
Y
Q
Q
770
|
Y
Y
M
M
D
D
D
D
L
L
Y
Y
V
V
G
G
S
S
D
D
780
|
L
L
E
E
I
I
G
G
Q
Q
H
H
R
R
T
T
K
K
I
I
790
|
E
E
E
E
L
L
R
R
Q
E
H
H
L
L
L
L
R
R
W
W
800
|
G
G
L
L
T
T
T
T
P
P
D
D
K
K
K
K
H
H
Q
Q
810
|
K
K
E
E
P
P
P
P
F
F
L
L
W
W
M
M
G
G
Y
Y
820
|
E
E
L
L
H
H
P
P
D
D
K
K
W
W
T
T
V
V
Q
Q
830
|
P
P
I
I
V
V
L
L
P
P
E
E
K
K
D
D
S
S
W
W
840
|
T
T
V
V
N
N
D
D
I
I
Q
Q
K
K
L
L
V
V
G
G
850
|
K
K
L
L
N
N
W
W
A
A
S
S
Q
Q
I
I
Y
Y
P
P
860
|
G
G
I
I
K
K
V
V
R
R
Q
Q
L
L
C
C
K
K
L
L
870
|
L
L
R
R
G
G
T
T
K
K
A
A
L
L
T
T
E
E
V
V
880
|
I
V
P
P
L
L
T
T
E
A
E
E
A
A
E
E
L
M
E
E
890
|
L
L
A
A
E
E
N
N
R
R
E
E
I
I
L
L
K
K
E
E
900
|
P
P
V
V
H
H
G
G
V
V
Y
Y
Y
Y
D
D
P
P
S
S
910
|
K
K
D
D
L
L
I
I
A
A
E
E
I
I
Q
Q
K
K
Q
Q
920
|
G
G
Q
Q
G
G
Q
Q
W
W
T
T
Y
Y
Q
Q
I
I
Y
Y
930
|
Q
Q
E
E
P
P
F
F
K
K
N
N
L
L
K
K
T
T
G
G
940
|
K
K
Y
Y
A
A
R
R
M
M
R
R
G
G
A
A
H
H
T
T
950
|
N
N
D
D
V
V
K
K
Q
Q
L
L
T
T
E
E
A
A
V
V
960
|
Q
Q
K
K
I
I
T
T
T
T
E
E
S
S
I
I
V
V
I
I
970
|
W
W
G
G
K
K
T
T
P
P
K
K
F
F
K
K
L
L
P
P
980
|
I
I
Q
Q
K
K
E
E
T
T
W
W
E
E
T
T
W
W
W
W
990
|
T
T
E
E
Y
Y
W
W
Q
Q
A
A
T
T
W
W
I
I
P
P
1000
|
E
E
W
W
E
E
F
F
V
V
N
N
T
T
P
P
P
P
L
L
1010
|
V
V
K
K
L
L
W
W
Y
Y
Q
Q
L
L
E
E
K
K
E
E
1020
|
P
P
I
I
V
V
G
G
A
A
E
E
T
T
F
F
Y
Y
V
V
1030
|
D
D
G
G
A
A
A
A
N
N
R
R
E
E
T
T
K
K
L
L
1040
|
G
G
K
K
A
A
G
G
Y
Y
V
V
T
T
N
N
R
R
G
G
1050
|
R
R
Q
Q
K
K
V
V
V
V
T
T
L
L
T
T
D
D
T
T
1060
|
T
T
N
N
Q
Q
K
K
T
T
E
E
L
L
Q
Q
A
A
I
I
1070
|
Y
Y
L
L
A
A
L
L
Q
Q
D
D
S
S
G
G
L
L
E
E
1080
|
V
V
N
N
I
I
V
V
T
T
D
D
S
S
Q
Q
Y
Y
A
A
1090
|
L
L
G
G
I
I
I
I
Q
Q
A
A
Q
Q
P
P
D
D
Q
Q
1100
|
S
S
E
E
S
S
E
E
L
L
V
V
N
N
Q
Q
I
I
I
I
1110
|
E
E
Q
Q
L
L
I
I
K
K
K
K
E
E
K
K
V
V
Y
Y
1120
|
L
L
A
A
W
W
V
V
P
P
A
A
H
H
K
K
G
G
I
I
1130
|
G
G
G
G
N
N
E
E
Q
Q
V
V
D
D
K
K
L
L
V
V
1140
|
S
S
A
A
G
G
I
I
R
R
K
K
V
V
L
L
|
|||||||||||||
| Experimental Note | Identified from the Human Clinical Data | ||||||||||||
| Experiment for Molecule Alteration |
Site-directed mutagenesis; Sanger sequencing assay | ||||||||||||
| Experiment for Drug Resistance |
PhenoSense assay | ||||||||||||
| Mechanism Description | Sixteen mutations at eight positions had a mean regression coefficient >=1.0 log10 (a contribution to decreased susceptibility of 10-fold or greater) for nevirapine, efavirenz, etravirine and/or rilpivirine: L100I, k101P, k103N/S, V106A/M, Y181C/I/V, Y188C/L, G190A/E/Q/S and F227C. With the exception of L100I and F227C, each mutation had a mean regression coefficient >=1.0 log10 for nevirapine. With the exception of k103S, V106A, Y181C/I/V and F227C, each had a mean regression coefficient >=1.0 log10 for efavirenz. Five mutations, k101P, Y181I/V, G190E and F227C, had a mean regression coefficient >=1.0 log10 for etravirine. Six mutations, L100I, k101P, Y181I/V, G190E and F227C, had a mean regression coefficient >= 1.0 log10 for rilpivirine. | ||||||||||||
| Key Molecule: HIV1 Reverse transcriptase (HIV1 RT) | [2] | ||||||||||||
| Sensitive Disease | Human immunodeficiency virus infection [ICD-11: 1C62.0] | ||||||||||||
| Molecule Alteration | Missense mutation | p.E6D+p.M41L+p.E44D+p.K49R+p.V60I+p.D67N+p.K70R+p.K103N+p.V108VI+p.D123E+p.I135T+p.M184V+p.T200I+p.I202V+p.T215V+p.K219E+p.P225PH+p.L228H+p.V245T+p.E248K+p.K249KR+p.I293V+p.E297A |
|||||||||||
| Wild Type Structure | Method: X-ray diffraction | Resolution: 2.09 Å | |||||||||||
| Mutant Type Structure | Method: AlphaFold | Average pLDDT: 87.004 |

|
||||||||||
| Download The Information of Sequence | Download The Structure File | ||||||||||||
-
P
P
I
I
590
|
S
S
P
P
I
I
E
D
T
T
V
V
P
P
V
V
K
K
L
L
600
|
K
K
P
P
G
G
M
M
D
D
G
G
P
P
K
K
V
V
K
K
610
|
Q
Q
W
W
P
P
L
L
T
T
E
E
E
E
K
K
I
I
K
K
620
|
A
A
L
L
V
V
E
E
I
I
C
C
T
T
E
E
M
L
E
E
630
|
K
K
E
D
G
G
K
K
I
I
S
S
K
R
I
I
G
G
P
P
640
|
E
E
N
N
P
P
Y
Y
N
N
T
T
P
P
V
I
F
F
A
A
650
|
I
I
K
K
K
K
K
K
D
N
S
S
T
T
K
R
W
W
R
R
660
|
K
K
L
L
V
V
D
D
F
F
R
R
E
E
L
L
N
N
K
K
670
|
R
R
T
T
Q
Q
D
D
F
F
W
W
E
E
V
V
Q
Q
L
L
680
|
G
G
I
I
P
P
H
H
P
P
A
A
G
G
L
L
K
K
K
K
690
|
K
N
K
K
S
S
V
V
T
T
V
I
L
L
D
D
V
V
G
G
700
|
D
D
A
A
Y
Y
F
F
S
S
V
V
P
P
L
L
D
D
E
E
710
|
D
E
F
F
R
R
K
K
Y
Y
T
T
A
A
F
F
T
T
I
I
720
|
P
P
S
S
I
T
N
N
N
N
E
E
T
T
P
P
G
G
I
I
730
|
R
R
Y
Y
Q
Q
Y
Y
N
N
V
V
L
L
P
P
Q
Q
G
G
740
|
W
W
K
K
G
G
S
S
P
P
A
A
I
I
F
F
Q
Q
S
S
750
|
S
S
M
M
T
T
K
K
I
I
L
L
E
E
P
P
F
F
R
R
760
|
K
K
Q
Q
N
N
P
P
D
D
I
I
V
V
I
I
Y
Y
Q
Q
770
|
Y
Y
M
V
D
D
D
D
L
L
Y
Y
V
V
G
G
S
S
D
D
780
|
L
L
E
E
I
I
G
G
Q
Q
H
H
R
R
T
I
K
K
I
V
790
|
E
E
E
E
L
L
R
R
Q
Q
H
H
L
L
L
L
R
R
W
W
800
|
G
G
L
L
T
V
T
T
P
P
D
D
K
E
K
K
H
H
Q
Q
810
|
K
K
E
E
P
H
P
P
F
F
L
H
W
W
M
M
G
G
Y
Y
820
|
E
E
L
L
H
H
P
P
D
D
K
K
W
W
T
T
V
V
Q
Q
830
|
P
P
I
I
V
T
L
L
P
P
E
K
K
R
D
D
S
S
W
W
840
|
T
T
V
V
N
N
D
D
I
I
Q
Q
K
K
L
L
V
V
G
G
850
|
K
K
L
L
N
N
W
W
A
A
S
S
Q
Q
I
I
Y
Y
P
P
860
|
G
G
I
I
K
K
V
V
R
R
Q
Q
L
L
C
C
K
K
L
L
870
|
L
L
R
R
G
G
T
T
K
K
A
A
L
L
T
T
E
E
V
V
880
|
I
V
P
P
L
L
T
T
E
A
E
E
A
A
E
E
L
L
E
E
890
|
L
L
A
A
E
E
N
N
R
R
E
E
I
I
L
L
K
K
E
E
900
|
P
P
V
V
H
H
G
G
V
V
Y
Y
Y
Y
D
D
P
P
S
S
910
|
K
K
D
D
L
L
I
I
A
A
E
E
I
I
Q
Q
K
K
Q
Q
920
|
G
G
Q
Q
G
G
Q
Q
W
W
T
T
Y
Y
Q
Q
I
I
Y
Y
930
|
Q
Q
E
E
P
P
F
F
K
K
N
N
L
L
K
K
T
T
G
G
940
|
K
K
Y
Y
A
A
R
R
M
M
R
R
G
G
A
A
H
H
T
T
950
|
N
N
D
D
V
V
K
K
Q
Q
L
L
T
T
E
E
A
A
V
V
960
|
Q
Q
K
K
I
I
T
T
T
T
E
E
S
S
I
I
V
V
I
I
970
|
W
W
G
G
K
K
T
T
P
P
K
K
F
F
K
K
L
L
P
P
980
|
I
I
Q
Q
K
K
E
E
T
T
W
W
E
E
T
T
W
W
W
W
990
|
T
T
E
E
Y
Y
W
W
Q
Q
A
A
T
T
W
W
I
I
P
P
1000
|
E
E
W
W
E
E
F
F
V
V
N
N
T
T
P
P
P
P
L
L
1010
|
V
V
K
K
L
L
W
W
Y
Y
Q
Q
L
L
E
E
K
K
E
E
1020
|
P
P
I
I
V
V
G
G
A
A
E
E
T
T
F
F
Y
Y
V
V
1030
|
D
D
G
G
A
A
A
A
N
N
R
R
E
E
T
T
K
K
L
L
1040
|
G
G
K
K
A
A
G
G
Y
Y
V
V
T
T
N
N
R
R
G
G
1050
|
R
R
Q
Q
K
K
V
V
V
V
T
T
L
L
T
T
D
D
T
T
1060
|
T
T
N
N
Q
Q
K
K
T
T
E
E
L
L
Q
Q
A
A
I
I
1070
|
Y
Y
L
L
A
A
L
L
Q
Q
D
D
S
S
G
G
L
L
E
E
1080
|
V
V
N
N
I
I
V
V
T
T
D
D
S
S
Q
Q
Y
Y
A
A
1090
|
L
L
G
G
I
I
I
I
Q
Q
A
A
Q
Q
P
P
D
D
Q
Q
1100
|
S
S
E
E
S
S
E
E
L
L
V
V
N
N
Q
Q
I
I
I
I
1110
|
E
E
Q
Q
L
L
I
I
K
K
K
K
E
E
K
K
V
V
Y
Y
1120
|
L
L
A
A
W
W
V
V
P
P
A
A
H
H
K
K
G
G
I
I
1130
|
G
G
G
G
N
N
E
E
Q
Q
V
V
D
D
K
K
L
L
V
V
1140
|
S
S
A
A
G
G
I
I
R
R
K
K
V
V
L
L
|
|||||||||||||
| Experimental Note | Identified from the Human Clinical Data | ||||||||||||
| Experiment for Molecule Alteration |
Site-directed mutagenesis; Sanger sequencing assay | ||||||||||||
| Experiment for Drug Resistance |
PhenoSense assay | ||||||||||||
| Mechanism Description | Sixteen mutations at eight positions had a mean regression coefficient >=1.0 log10 (a contribution to decreased susceptibility of 10-fold or greater) for nevirapine, efavirenz, etravirine and/or rilpivirine: L100I, k101P, k103N/S, V106A/M, Y181C/I/V, Y188C/L, G190A/E/Q/S and F227C. With the exception of L100I and F227C, each mutation had a mean regression coefficient >=1.0 log10 for nevirapine. With the exception of k103S, V106A, Y181C/I/V and F227C, each had a mean regression coefficient >=1.0 log10 for efavirenz. Five mutations, k101P, Y181I/V, G190E and F227C, had a mean regression coefficient >=1.0 log10 for etravirine. Six mutations, L100I, k101P, Y181I/V, G190E and F227C, had a mean regression coefficient >= 1.0 log10 for rilpivirine. | ||||||||||||
| Key Molecule: HIV1 Reverse transcriptase (HIV1 RT) | [2] | ||||||||||||
| Sensitive Disease | Human immunodeficiency virus infection [ICD-11: 1C62.0] | ||||||||||||
| Molecule Alteration | Missense mutation | p.V90I+p.D123E+p.I202V+p.E204Q+p.Q207D+p.R211K+p.V245T+p.I293V+p.E297A |
|||||||||||
| Wild Type Structure | Method: X-ray diffraction | Resolution: 2.09 Å | |||||||||||
| Mutant Type Structure | Method: AlphaFold | Average pLDDT: 86.863 |

|
||||||||||
| Download The Information of Sequence | Download The Structure File | ||||||||||||
-
P
P
I
I
590
|
S
S
P
P
I
I
E
E
T
T
V
V
P
P
V
V
K
K
L
L
600
|
K
K
P
P
G
G
M
M
D
D
G
G
P
P
K
K
V
V
K
K
610
|
Q
Q
W
W
P
P
L
L
T
T
E
E
E
E
K
K
I
I
K
K
620
|
A
A
L
L
V
V
E
E
I
I
C
C
T
T
E
E
M
M
E
E
630
|
K
K
E
E
G
G
K
K
I
I
S
S
K
K
I
I
G
G
P
P
640
|
E
E
N
N
P
P
Y
Y
N
N
T
T
P
P
V
V
F
F
A
A
650
|
I
I
K
K
K
K
K
K
D
D
S
S
T
T
K
K
W
W
R
R
660
|
K
K
L
L
V
V
D
D
F
F
R
R
E
E
L
L
N
N
K
K
670
|
R
R
T
T
Q
Q
D
D
F
F
W
W
E
E
V
I
Q
Q
L
L
680
|
G
G
I
I
P
P
H
H
P
P
A
A
G
G
L
L
K
K
K
K
690
|
K
K
K
K
S
S
V
V
T
T
V
V
L
L
D
D
V
V
G
G
700
|
D
D
A
A
Y
Y
F
F
S
S
V
V
P
P
L
L
D
D
E
E
710
|
D
E
F
F
R
R
K
K
Y
Y
T
T
A
A
F
F
T
T
I
I
720
|
P
P
S
S
I
I
N
N
N
N
E
E
T
T
P
P
G
G
I
I
730
|
R
R
Y
Y
Q
Q
Y
Y
N
N
V
V
L
L
P
P
Q
Q
G
G
740
|
W
W
K
K
G
G
S
S
P
P
A
A
I
I
F
F
Q
Q
S
S
750
|
S
S
M
M
T
T
K
K
I
I
L
L
E
E
P
P
F
F
R
R
760
|
K
K
Q
Q
N
N
P
P
D
D
I
I
V
V
I
I
Y
Y
Q
Q
770
|
Y
Y
M
M
D
D
D
D
L
L
Y
Y
V
V
G
G
S
S
D
D
780
|
L
L
E
E
I
I
G
G
Q
Q
H
H
R
R
T
T
K
K
I
V
790
|
E
E
E
Q
L
L
R
R
Q
D
H
H
L
L
L
L
R
K
W
W
800
|
G
G
L
L
T
T
T
T
P
P
D
D
K
K
K
K
H
H
Q
Q
810
|
K
K
E
E
P
P
P
P
F
F
L
L
W
W
M
M
G
G
Y
Y
820
|
E
E
L
L
H
H
P
P
D
D
K
K
W
W
T
T
V
V
Q
Q
830
|
P
P
I
I
V
T
L
L
P
P
E
E
K
K
D
D
S
S
W
W
840
|
T
T
V
V
N
N
D
D
I
I
Q
Q
K
K
L
L
V
V
G
G
850
|
K
K
L
L
N
N
W
W
A
A
S
S
Q
Q
I
I
Y
Y
P
P
860
|
G
G
I
I
K
K
V
V
R
R
Q
Q
L
L
C
C
K
K
L
L
870
|
L
L
R
R
G
G
T
T
K
K
A
A
L
L
T
T
E
E
V
V
880
|
I
V
P
P
L
L
T
T
E
A
E
E
A
A
E
E
L
L
E
E
890
|
L
L
A
A
E
E
N
N
R
R
E
E
I
I
L
L
K
K
E
E
900
|
P
P
V
V
H
H
G
G
V
V
Y
Y
Y
Y
D
D
P
P
S
S
910
|
K
K
D
D
L
L
I
I
A
A
E
E
I
I
Q
Q
K
K
Q
Q
920
|
G
G
Q
Q
G
G
Q
Q
W
W
T
T
Y
Y
Q
Q
I
I
Y
Y
930
|
Q
Q
E
E
P
P
F
F
K
K
N
N
L
L
K
K
T
T
G
G
940
|
K
K
Y
Y
A
A
R
R
M
M
R
R
G
G
A
A
H
H
T
T
950
|
N
N
D
D
V
V
K
K
Q
Q
L
L
T
T
E
E
A
A
V
V
960
|
Q
Q
K
K
I
I
T
T
T
T
E
E
S
S
I
I
V
V
I
I
970
|
W
W
G
G
K
K
T
T
P
P
K
K
F
F
K
K
L
L
P
P
980
|
I
I
Q
Q
K
K
E
E
T
T
W
W
E
E
T
T
W
W
W
W
990
|
T
T
E
E
Y
Y
W
W
Q
Q
A
A
T
T
W
W
I
I
P
P
1000
|
E
E
W
W
E
E
F
F
V
V
N
N
T
T
P
P
P
P
L
L
1010
|
V
V
K
K
L
L
W
W
Y
Y
Q
Q
L
L
E
E
K
K
E
E
1020
|
P
P
I
I
V
V
G
G
A
A
E
E
T
T
F
F
Y
Y
V
V
1030
|
D
D
G
G
A
A
A
A
N
N
R
R
E
E
T
T
K
K
L
L
1040
|
G
G
K
K
A
A
G
G
Y
Y
V
V
T
T
N
N
R
R
G
G
1050
|
R
R
Q
Q
K
K
V
V
V
V
T
T
L
L
T
T
D
D
T
T
1060
|
T
T
N
N
Q
Q
K
K
T
T
E
E
L
L
Q
Q
A
A
I
I
1070
|
Y
Y
L
L
A
A
L
L
Q
Q
D
D
S
S
G
G
L
L
E
E
1080
|
V
V
N
N
I
I
V
V
T
T
D
D
S
S
Q
Q
Y
Y
A
A
1090
|
L
L
G
G
I
I
I
I
Q
Q
A
A
Q
Q
P
P
D
D
Q
Q
1100
|
S
S
E
E
S
S
E
E
L
L
V
V
N
N
Q
Q
I
I
I
I
1110
|
E
E
Q
Q
L
L
I
I
K
K
K
K
E
E
K
K
V
V
Y
Y
1120
|
L
L
A
A
W
W
V
V
P
P
A
A
H
H
K
K
G
G
I
I
1130
|
G
G
G
G
N
N
E
E
Q
Q
V
V
D
D
K
K
L
L
V
V
1140
|
S
S
A
A
G
G
I
I
R
R
K
K
V
V
L
L
|
|||||||||||||
| Experimental Note | Identified from the Human Clinical Data | ||||||||||||
| Experiment for Molecule Alteration |
Site-directed mutagenesis; Sanger sequencing assay | ||||||||||||
| Experiment for Drug Resistance |
PhenoSense assay | ||||||||||||
| Mechanism Description | Sixteen mutations at eight positions had a mean regression coefficient >=1.0 log10 (a contribution to decreased susceptibility of 10-fold or greater) for nevirapine, efavirenz, etravirine and/or rilpivirine: L100I, k101P, k103N/S, V106A/M, Y181C/I/V, Y188C/L, G190A/E/Q/S and F227C. With the exception of L100I and F227C, each mutation had a mean regression coefficient >=1.0 log10 for nevirapine. With the exception of k103S, V106A, Y181C/I/V and F227C, each had a mean regression coefficient >=1.0 log10 for efavirenz. Five mutations, k101P, Y181I/V, G190E and F227C, had a mean regression coefficient >=1.0 log10 for etravirine. Six mutations, L100I, k101P, Y181I/V, G190E and F227C, had a mean regression coefficient >= 1.0 log10 for rilpivirine. | ||||||||||||
| Key Molecule: HIV1 Reverse transcriptase (HIV1 RT) | [2] | ||||||||||||
| Sensitive Disease | Human immunodeficiency virus infection [ICD-11: 1C62.0] | ||||||||||||
| Molecule Alteration | Missense mutation | p.P4PH+p.K20R+p.T39A+p.L74LI+p.R83K+p.V106VI+p.V179VI+p.M184V+p.E224D+p.V245E+p.T286A+p.P294PS+p.E297A |
|||||||||||
| Wild Type Structure | Method: X-ray diffraction | Resolution: 2.09 Å | |||||||||||
| Mutant Type Structure | Method: AlphaFold | Average pLDDT: 87.637 |

|
||||||||||
| Download The Information of Sequence | Download The Structure File | ||||||||||||
-
P
P
I
I
590
|
S
S
P
H
I
I
E
E
T
T
V
V
P
P
V
V
K
K
L
L
600
|
K
K
P
P
G
G
M
M
D
D
G
G
P
P
K
R
V
V
K
K
610
|
Q
Q
W
W
P
P
L
L
T
T
E
E
E
E
K
K
I
I
K
K
620
|
A
A
L
L
V
V
E
E
I
I
C
C
T
A
E
E
M
M
E
E
630
|
K
K
E
E
G
G
K
K
I
I
S
S
K
K
I
I
G
G
P
P
640
|
E
E
N
N
P
P
Y
Y
N
N
T
T
P
P
V
V
F
F
A
A
650
|
I
I
K
K
K
K
K
K
D
D
S
S
T
T
K
K
W
W
R
R
660
|
K
K
L
I
V
V
D
D
F
F
R
R
E
E
L
L
N
N
K
K
670
|
R
K
T
T
Q
Q
D
D
F
F
W
W
E
E
V
V
Q
Q
L
L
680
|
G
G
I
I
P
P
H
H
P
P
A
A
G
G
L
L
K
K
K
K
690
|
K
K
K
K
S
S
V
I
T
T
V
V
L
L
D
D
V
V
G
G
700
|
D
D
A
A
Y
Y
F
F
S
S
V
V
P
P
L
L
D
D
E
E
710
|
D
D
F
F
R
R
K
K
Y
Y
T
T
A
A
F
F
T
T
I
I
720
|
P
P
S
S
I
I
N
N
N
N
E
E
T
T
P
P
G
G
I
I
730
|
R
R
Y
Y
Q
Q
Y
Y
N
N
V
V
L
L
P
P
Q
Q
G
G
740
|
W
W
K
K
G
G
S
S
P
P
A
A
I
I
F
F
Q
Q
S
S
750
|
S
S
M
M
T
T
K
K
I
I
L
L
E
E
P
P
F
F
R
R
760
|
K
K
Q
Q
N
N
P
P
D
D
I
I
V
I
I
I
Y
Y
Q
Q
770
|
Y
Y
M
V
D
D
D
D
L
L
Y
Y
V
V
G
G
S
S
D
D
780
|
L
L
E
E
I
I
G
G
Q
Q
H
H
R
R
T
T
K
K
I
I
790
|
E
E
E
E
L
L
R
R
Q
Q
H
H
L
L
L
L
R
R
W
W
800
|
G
G
L
L
T
T
T
T
P
P
D
D
K
K
K
K
H
H
Q
Q
810
|
K
K
E
D
P
P
P
P
F
F
L
L
W
W
M
M
G
G
Y
Y
820
|
E
E
L
L
H
H
P
P
D
D
K
K
W
W
T
T
V
V
Q
Q
830
|
P
P
I
I
V
E
L
L
P
P
E
E
K
K
D
D
S
S
W
W
840
|
T
T
V
V
N
N
D
D
I
I
Q
Q
K
K
L
L
V
V
G
G
850
|
K
K
L
L
N
N
W
W
A
A
S
S
Q
Q
I
I
Y
Y
P
P
860
|
G
G
I
I
K
K
V
V
R
R
Q
Q
L
L
C
C
K
K
L
L
870
|
L
L
R
R
G
G
T
A
K
K
A
A
L
L
T
T
E
E
V
V
880
|
I
I
P
S
L
L
T
T
E
A
E
E
A
A
E
E
L
L
E
E
890
|
L
L
A
A
E
E
N
N
R
R
E
E
I
I
L
L
K
K
E
E
900
|
P
P
V
V
H
H
G
G
V
V
Y
Y
Y
Y
D
D
P
P
S
S
910
|
K
K
D
D
L
L
I
I
A
A
E
E
I
I
Q
Q
K
K
Q
Q
920
|
G
G
Q
Q
G
G
Q
Q
W
W
T
T
Y
Y
Q
Q
I
I
Y
Y
930
|
Q
Q
E
E
P
P
F
F
K
K
N
N
L
L
K
K
T
T
G
G
940
|
K
K
Y
Y
A
A
R
R
M
M
R
R
G
G
A
A
H
H
T
T
950
|
N
N
D
D
V
V
K
K
Q
Q
L
L
T
T
E
E
A
A
V
V
960
|
Q
Q
K
K
I
I
T
T
T
T
E
E
S
S
I
I
V
V
I
I
970
|
W
W
G
G
K
K
T
T
P
P
K
K
F
F
K
K
L
L
P
P
980
|
I
I
Q
Q
K
K
E
E
T
T
W
W
E
E
T
T
W
W
W
W
990
|
T
T
E
E
Y
Y
W
W
Q
Q
A
A
T
T
W
W
I
I
P
P
1000
|
E
E
W
W
E
E
F
F
V
V
N
N
T
T
P
P
P
P
L
L
1010
|
V
V
K
K
L
L
W
W
Y
Y
Q
Q
L
L
E
E
K
K
E
E
1020
|
P
P
I
I
V
V
G
G
A
A
E
E
T
T
F
F
Y
Y
V
V
1030
|
D
D
G
G
A
A
A
A
N
N
R
R
E
E
T
T
K
K
L
L
1040
|
G
G
K
K
A
A
G
G
Y
Y
V
V
T
T
N
N
R
R
G
G
1050
|
R
R
Q
Q
K
K
V
V
V
V
T
T
L
L
T
T
D
D
T
T
1060
|
T
T
N
N
Q
Q
K
K
T
T
E
E
L
L
Q
Q
A
A
I
I
1070
|
Y
Y
L
L
A
A
L
L
Q
Q
D
D
S
S
G
G
L
L
E
E
1080
|
V
V
N
N
I
I
V
V
T
T
D
D
S
S
Q
Q
Y
Y
A
A
1090
|
L
L
G
G
I
I
I
I
Q
Q
A
A
Q
Q
P
P
D
D
Q
Q
1100
|
S
S
E
E
S
S
E
E
L
L
V
V
N
N
Q
Q
I
I
I
I
1110
|
E
E
Q
Q
L
L
I
I
K
K
K
K
E
E
K
K
V
V
Y
Y
1120
|
L
L
A
A
W
W
V
V
P
P
A
A
H
H
K
K
G
G
I
I
1130
|
G
G
G
G
N
N
E
E
Q
Q
V
V
D
D
K
K
L
L
V
V
1140
|
S
S
A
A
G
G
I
I
R
R
K
K
V
V
L
L
|
|||||||||||||
| Experimental Note | Identified from the Human Clinical Data | ||||||||||||
| Experiment for Molecule Alteration |
Site-directed mutagenesis; Sanger sequencing assay | ||||||||||||
| Experiment for Drug Resistance |
PhenoSense assay | ||||||||||||
| Mechanism Description | Sixteen mutations at eight positions had a mean regression coefficient >=1.0 log10 (a contribution to decreased susceptibility of 10-fold or greater) for nevirapine, efavirenz, etravirine and/or rilpivirine: L100I, k101P, k103N/S, V106A/M, Y181C/I/V, Y188C/L, G190A/E/Q/S and F227C. With the exception of L100I and F227C, each mutation had a mean regression coefficient >=1.0 log10 for nevirapine. With the exception of k103S, V106A, Y181C/I/V and F227C, each had a mean regression coefficient >=1.0 log10 for efavirenz. Five mutations, k101P, Y181I/V, G190E and F227C, had a mean regression coefficient >=1.0 log10 for etravirine. Six mutations, L100I, k101P, Y181I/V, G190E and F227C, had a mean regression coefficient >= 1.0 log10 for rilpivirine. | ||||||||||||
| Key Molecule: HIV1 Reverse transcriptase (HIV1 RT) | [2] | ||||||||||||
| Sensitive Disease | Human immunodeficiency virus infection [ICD-11: 1C62.0] | ||||||||||||
| Molecule Alteration | Missense mutation | p.D67N+p.K70R+p.I135T+p.S162C+p.Q174QE+p.Y181C+p.M184V+p.Q207KR+p.R211Q+p.K219Q+p.H221Y+p.I293V |
|||||||||||
| Wild Type Structure | Method: X-ray diffraction | Resolution: 2.09 Å | |||||||||||
| Mutant Type Structure | Method: AlphaFold | Average pLDDT: 86.816 |

|
||||||||||
| Download The Information of Sequence | Download The Structure File | ||||||||||||
-
P
P
I
I
590
|
S
S
P
P
I
I
E
E
T
T
V
V
P
P
V
V
K
K
L
L
600
|
K
K
P
P
G
G
M
M
D
D
G
G
P
P
K
K
V
V
K
K
610
|
Q
Q
W
W
P
P
L
L
T
T
E
E
E
E
K
K
I
I
K
K
620
|
A
A
L
L
V
V
E
E
I
I
C
C
T
T
E
E
M
M
E
E
630
|
K
K
E
E
G
G
K
K
I
I
S
S
K
K
I
I
G
G
P
P
640
|
E
E
N
N
P
P
Y
Y
N
N
T
T
P
P
V
V
F
F
A
A
650
|
I
I
K
K
K
K
K
K
D
N
S
S
T
T
K
R
W
W
R
R
660
|
K
K
L
L
V
V
D
D
F
F
R
R
E
E
L
L
N
N
K
K
670
|
R
R
T
T
Q
Q
D
D
F
F
W
W
E
E
V
V
Q
Q
L
L
680
|
G
G
I
I
P
P
H
H
P
P
A
A
G
G
L
L
K
K
K
K
690
|
K
K
K
K
S
S
V
V
T
T
V
V
L
L
D
D
V
V
G
G
700
|
D
D
A
A
Y
Y
F
F
S
S
V
V
P
P
L
L
D
D
E
E
710
|
D
D
F
F
R
R
K
K
Y
Y
T
T
A
A
F
F
T
T
I
I
720
|
P
P
S
S
I
T
N
N
N
N
E
E
T
T
P
P
G
G
I
I
730
|
R
R
Y
Y
Q
Q
Y
Y
N
N
V
V
L
L
P
P
Q
Q
G
G
740
|
W
W
K
K
G
G
S
S
P
P
A
A
I
I
F
F
Q
Q
S
C
750
|
S
S
M
M
T
T
K
K
I
I
L
L
E
E
P
P
F
F
R
R
760
|
K
K
Q
E
N
N
P
P
D
D
I
I
V
V
I
I
Y
C
Q
Q
770
|
Y
Y
M
V
D
D
D
D
L
L
Y
Y
V
V
G
G
S
S
D
D
780
|
L
L
E
E
I
I
G
G
Q
Q
H
H
R
R
T
T
K
K
I
I
790
|
E
E
E
E
L
L
R
R
Q
K
H
H
L
L
L
L
R
Q
W
W
800
|
G
G
L
L
T
T
T
T
P
P
D
D
K
Q
K
K
H
Y
Q
Q
810
|
K
K
E
E
P
P
P
P
F
F
L
L
W
W
M
M
G
G
Y
Y
820
|
E
E
L
L
H
H
P
P
D
D
K
K
W
W
T
T
V
V
Q
Q
830
|
P
P
I
I
V
V
L
L
P
P
E
E
K
K
D
D
S
S
W
W
840
|
T
T
V
V
N
N
D
D
I
I
Q
Q
K
K
L
L
V
V
G
G
850
|
K
K
L
L
N
N
W
W
A
A
S
S
Q
Q
I
I
Y
Y
P
P
860
|
G
G
I
I
K
K
V
V
R
R
Q
Q
L
L
C
C
K
K
L
L
870
|
L
L
R
R
G
G
T
T
K
K
A
A
L
L
T
T
E
E
V
V
880
|
I
V
P
P
L
L
T
T
E
E
E
E
A
A
E
E
L
L
E
E
890
|
L
L
A
A
E
E
N
N
R
R
E
E
I
I
L
L
K
K
E
E
900
|
P
P
V
V
H
H
G
G
V
V
Y
Y
Y
Y
D
D
P
P
S
S
910
|
K
K
D
D
L
L
I
I
A
A
E
E
I
I
Q
Q
K
K
Q
Q
920
|
G
G
Q
Q
G
G
Q
Q
W
W
T
T
Y
Y
Q
Q
I
I
Y
Y
930
|
Q
Q
E
E
P
P
F
F
K
K
N
N
L
L
K
K
T
T
G
G
940
|
K
K
Y
Y
A
A
R
R
M
M
R
R
G
G
A
A
H
H
T
T
950
|
N
N
D
D
V
V
K
K
Q
Q
L
L
T
T
E
E
A
A
V
V
960
|
Q
Q
K
K
I
I
T
T
T
T
E
E
S
S
I
I
V
V
I
I
970
|
W
W
G
G
K
K
T
T
P
P
K
K
F
F
K
K
L
L
P
P
980
|
I
I
Q
Q
K
K
E
E
T
T
W
W
E
E
T
T
W
W
W
W
990
|
T
T
E
E
Y
Y
W
W
Q
Q
A
A
T
T
W
W
I
I
P
P
1000
|
E
E
W
W
E
E
F
F
V
V
N
N
T
T
P
P
P
P
L
L
1010
|
V
V
K
K
L
L
W
W
Y
Y
Q
Q
L
L
E
E
K
K
E
E
1020
|
P
P
I
I
V
V
G
G
A
A
E
E
T
T
F
F
Y
Y
V
V
1030
|
D
D
G
G
A
A
A
A
N
N
R
R
E
E
T
T
K
K
L
L
1040
|
G
G
K
K
A
A
G
G
Y
Y
V
V
T
T
N
N
R
R
G
G
1050
|
R
R
Q
Q
K
K
V
V
V
V
T
T
L
L
T
T
D
D
T
T
1060
|
T
T
N
N
Q
Q
K
K
T
T
E
E
L
L
Q
Q
A
A
I
I
1070
|
Y
Y
L
L
A
A
L
L
Q
Q
D
D
S
S
G
G
L
L
E
E
1080
|
V
V
N
N
I
I
V
V
T
T
D
D
S
S
Q
Q
Y
Y
A
A
1090
|
L
L
G
G
I
I
I
I
Q
Q
A
A
Q
Q
P
P
D
D
Q
Q
1100
|
S
S
E
E
S
S
E
E
L
L
V
V
N
N
Q
Q
I
I
I
I
1110
|
E
E
Q
Q
L
L
I
I
K
K
K
K
E
E
K
K
V
V
Y
Y
1120
|
L
L
A
A
W
W
V
V
P
P
A
A
H
H
K
K
G
G
I
I
1130
|
G
G
G
G
N
N
E
E
Q
Q
V
V
D
D
K
K
L
L
V
V
1140
|
S
S
A
A
G
G
I
I
R
R
K
K
V
V
L
L
|
|||||||||||||
| Experimental Note | Identified from the Human Clinical Data | ||||||||||||
| Experiment for Molecule Alteration |
Site-directed mutagenesis; Sanger sequencing assay | ||||||||||||
| Experiment for Drug Resistance |
PhenoSense assay | ||||||||||||
| Mechanism Description | Sixteen mutations at eight positions had a mean regression coefficient >=1.0 log10 (a contribution to decreased susceptibility of 10-fold or greater) for nevirapine, efavirenz, etravirine and/or rilpivirine: L100I, k101P, k103N/S, V106A/M, Y181C/I/V, Y188C/L, G190A/E/Q/S and F227C. With the exception of L100I and F227C, each mutation had a mean regression coefficient >=1.0 log10 for nevirapine. With the exception of k103S, V106A, Y181C/I/V and F227C, each had a mean regression coefficient >=1.0 log10 for efavirenz. Five mutations, k101P, Y181I/V, G190E and F227C, had a mean regression coefficient >=1.0 log10 for etravirine. Six mutations, L100I, k101P, Y181I/V, G190E and F227C, had a mean regression coefficient >= 1.0 log10 for rilpivirine. | ||||||||||||
| Key Molecule: HIV1 Reverse transcriptase (HIV1 RT) | [2] | ||||||||||||
| Sensitive Disease | Human immunodeficiency virus infection [ICD-11: 1C62.0] | ||||||||||||
| Molecule Alteration | Missense mutation | p.E28K+p.S68G+p.A98S+p.K101E+p.D123E+p.I135V+p.I142IT+p.R143RG+p.M184V+p.L210LW+p.R211K+p.T215Y+p.T286TA+p.E297A |
|||||||||||
| Wild Type Structure | Method: X-ray diffraction | Resolution: 2.09 Å | |||||||||||
| Mutant Type Structure | Method: AlphaFold | Average pLDDT: 87.235 |

|
||||||||||
| Download The Information of Sequence | Download The Structure File | ||||||||||||
-
P
P
I
I
590
|
S
S
P
P
I
I
E
E
T
T
V
V
P
P
V
V
K
K
L
L
600
|
K
K
P
P
G
G
M
M
D
D
G
G
P
P
K
K
V
V
K
K
610
|
Q
Q
W
W
P
P
L
L
T
T
E
K
E
E
K
K
I
I
K
K
620
|
A
A
L
L
V
V
E
E
I
I
C
C
T
T
E
E
M
M
E
E
630
|
K
K
E
E
G
G
K
K
I
I
S
S
K
K
I
I
G
G
P
P
640
|
E
E
N
N
P
P
Y
Y
N
N
T
T
P
P
V
V
F
F
A
A
650
|
I
I
K
K
K
K
K
K
D
D
S
G
T
T
K
K
W
W
R
R
660
|
K
K
L
L
V
V
D
D
F
F
R
R
E
E
L
L
N
N
K
K
670
|
R
R
T
T
Q
Q
D
D
F
F
W
W
E
E
V
V
Q
Q
L
L
680
|
G
G
I
I
P
P
H
H
P
P
A
S
G
G
L
L
K
E
K
K
690
|
K
K
K
K
S
S
V
V
T
T
V
V
L
L
D
D
V
V
G
G
700
|
D
D
A
A
Y
Y
F
F
S
S
V
V
P
P
L
L
D
D
E
E
710
|
D
E
F
F
R
R
K
K
Y
Y
T
T
A
A
F
F
T
T
I
I
720
|
P
P
S
S
I
V
N
N
N
N
E
E
T
T
P
P
G
G
I
T
730
|
R
G
Y
Y
Q
Q
Y
Y
N
N
V
V
L
L
P
P
Q
Q
G
G
740
|
W
W
K
K
G
G
S
S
P
P
A
A
I
I
F
F
Q
Q
S
S
750
|
S
S
M
M
T
T
K
K
I
I
L
L
E
E
P
P
F
F
R
R
760
|
K
K
Q
Q
N
N
P
P
D
D
I
I
V
V
I
I
Y
Y
Q
Q
770
|
Y
Y
M
V
D
D
D
D
L
L
Y
Y
V
V
G
G
S
S
D
D
780
|
L
L
E
E
I
I
G
G
Q
Q
H
H
R
R
T
T
K
K
I
I
790
|
E
E
E
E
L
L
R
R
Q
Q
H
H
L
L
L
W
R
K
W
W
800
|
G
G
L
L
T
Y
T
T
P
P
D
D
K
K
K
K
H
H
Q
Q
810
|
K
K
E
E
P
P
P
P
F
F
L
L
W
W
M
M
G
G
Y
Y
820
|
E
E
L
L
H
H
P
P
D
D
K
K
W
W
T
T
V
V
Q
Q
830
|
P
P
I
I
V
V
L
L
P
P
E
E
K
K
D
D
S
S
W
W
840
|
T
T
V
V
N
N
D
D
I
I
Q
Q
K
K
L
L
V
V
G
G
850
|
K
K
L
L
N
N
W
W
A
A
S
S
Q
Q
I
I
Y
Y
P
P
860
|
G
G
I
I
K
K
V
V
R
R
Q
Q
L
L
C
C
K
K
L
L
870
|
L
L
R
R
G
G
T
A
K
K
A
A
L
L
T
T
E
E
V
V
880
|
I
I
P
P
L
L
T
T
E
A
E
E
A
A
E
E
L
L
E
E
890
|
L
L
A
A
E
E
N
N
R
R
E
E
I
I
L
L
K
K
E
E
900
|
P
P
V
V
H
H
G
G
V
V
Y
Y
Y
Y
D
D
P
P
S
S
910
|
K
K
D
D
L
L
I
I
A
A
E
E
I
I
Q
Q
K
K
Q
Q
920
|
G
G
Q
Q
G
G
Q
Q
W
W
T
T
Y
Y
Q
Q
I
I
Y
Y
930
|
Q
Q
E
E
P
P
F
F
K
K
N
N
L
L
K
K
T
T
G
G
940
|
K
K
Y
Y
A
A
R
R
M
M
R
R
G
G
A
A
H
H
T
T
950
|
N
N
D
D
V
V
K
K
Q
Q
L
L
T
T
E
E
A
A
V
V
960
|
Q
Q
K
K
I
I
T
T
T
T
E
E
S
S
I
I
V
V
I
I
970
|
W
W
G
G
K
K
T
T
P
P
K
K
F
F
K
K
L
L
P
P
980
|
I
I
Q
Q
K
K
E
E
T
T
W
W
E
E
T
T
W
W
W
W
990
|
T
T
E
E
Y
Y
W
W
Q
Q
A
A
T
T
W
W
I
I
P
P
1000
|
E
E
W
W
E
E
F
F
V
V
N
N
T
T
P
P
P
P
L
L
1010
|
V
V
K
K
L
L
W
W
Y
Y
Q
Q
L
L
E
E
K
K
E
E
1020
|
P
P
I
I
V
V
G
G
A
A
E
E
T
T
F
F
Y
Y
V
V
1030
|
D
D
G
G
A
A
A
A
N
N
R
R
E
E
T
T
K
K
L
L
1040
|
G
G
K
K
A
A
G
G
Y
Y
V
V
T
T
N
N
R
R
G
G
1050
|
R
R
Q
Q
K
K
V
V
V
V
T
T
L
L
T
T
D
D
T
T
1060
|
T
T
N
N
Q
Q
K
K
T
T
E
E
L
L
Q
Q
A
A
I
I
1070
|
Y
Y
L
L
A
A
L
L
Q
Q
D
D
S
S
G
G
L
L
E
E
1080
|
V
V
N
N
I
I
V
V
T
T
D
D
S
S
Q
Q
Y
Y
A
A
1090
|
L
L
G
G
I
I
I
I
Q
Q
A
A
Q
Q
P
P
D
D
Q
Q
1100
|
S
S
E
E
S
S
E
E
L
L
V
V
N
N
Q
Q
I
I
I
I
1110
|
E
E
Q
Q
L
L
I
I
K
K
K
K
E
E
K
K
V
V
Y
Y
1120
|
L
L
A
A
W
W
V
V
P
P
A
A
H
H
K
K
G
G
I
I
1130
|
G
G
G
G
N
N
E
E
Q
Q
V
V
D
D
K
K
L
L
V
V
1140
|
S
S
A
A
G
G
I
I
R
R
K
K
V
V
L
L
|
|||||||||||||
| Experimental Note | Identified from the Human Clinical Data | ||||||||||||
| Experiment for Molecule Alteration |
Site-directed mutagenesis; Sanger sequencing assay | ||||||||||||
| Experiment for Drug Resistance |
PhenoSense assay | ||||||||||||
| Mechanism Description | Sixteen mutations at eight positions had a mean regression coefficient >=1.0 log10 (a contribution to decreased susceptibility of 10-fold or greater) for nevirapine, efavirenz, etravirine and/or rilpivirine: L100I, k101P, k103N/S, V106A/M, Y181C/I/V, Y188C/L, G190A/E/Q/S and F227C. With the exception of L100I and F227C, each mutation had a mean regression coefficient >=1.0 log10 for nevirapine. With the exception of k103S, V106A, Y181C/I/V and F227C, each had a mean regression coefficient >=1.0 log10 for efavirenz. Five mutations, k101P, Y181I/V, G190E and F227C, had a mean regression coefficient >=1.0 log10 for etravirine. Six mutations, L100I, k101P, Y181I/V, G190E and F227C, had a mean regression coefficient >= 1.0 log10 for rilpivirine. | ||||||||||||
| Key Molecule: HIV1 Reverse transcriptase (HIV1 RT) | [3] | ||||||||||||
| Sensitive Disease | Human immunodeficiency virus infection [ICD-11: 1C62.0] | ||||||||||||
| Molecule Alteration | Missense mutation | p.V8VI+p.K20KR+p.V35I+p.D123E+p.I135T+p.K173T+p.Q174K+p.D177E+p.V179VD+p.G196E+p.T200K+p.Q207A+p.P243T+p.V245K+p.D256E+p.A272AP+p.T286TA+p.E291D+p.V292I+p.I293V+p.E297A |
|||||||||||
| Experimental Note | Identified from the Human Clinical Data | ||||||||||||
| Experiment for Molecule Alteration |
GeneSeq assay; PhenoSense GT assay | ||||||||||||
| Experiment for Drug Resistance |
Viral load assay | ||||||||||||
| Mechanism Description | Amino acid changes in IN may contribute to drug resistance or sensitivity. | ||||||||||||
| Key Molecule: HIV1 Reverse transcriptase (HIV1 RT) | [3] | ||||||||||||
| Sensitive Disease | Human immunodeficiency virus infection [ICD-11: 1C62.0] | ||||||||||||
| Molecule Alteration | Missense mutation | p.T39S+p.S48T+p.V60I+p.S68G+p.K102Q+p.K122KE+p.D123X+p.A158S+p.I178L+p.V179DE+p.I195IL+p.T200TA+p.R211K+p.I244V+p.V245E+p.A272P+p.K277KR+p.R284K+p.A288S+p.V292I+p.I293V+p.E297A |
|||||||||||
| Experimental Note | Identified from the Human Clinical Data | ||||||||||||
| Experiment for Molecule Alteration |
GeneSeq assay; PhenoSense GT assay | ||||||||||||
| Experiment for Drug Resistance |
Viral load assay | ||||||||||||
| Mechanism Description | Amino acid changes in IN may contribute to drug resistance or sensitivity. | ||||||||||||
| Key Molecule: HIV1 Reverse transcriptase (HIV1 RT) | [3] | ||||||||||||
| Sensitive Disease | Human immunodeficiency virus infection [ICD-11: 1C62.0] | ||||||||||||
| Molecule Alteration | Missense mutation | p.V35E+p.T39A+p.D177E+p.G196E+p.Q207E+p.R211K+p.V245K+p.I257L+p.K277R+p.T286A+p.I293V |
|||||||||||
| Experimental Note | Identified from the Human Clinical Data | ||||||||||||
| Experiment for Molecule Alteration |
GeneSeq assay; PhenoSense GT assay | ||||||||||||
| Experiment for Drug Resistance |
Viral load assay | ||||||||||||
| Mechanism Description | Amino acid changes in IN may contribute to drug resistance or sensitivity. | ||||||||||||
| Key Molecule: HIV1 Reverse transcriptase (HIV1 RT) | [3] | ||||||||||||
| Sensitive Disease | Human immunodeficiency virus infection [ICD-11: 1C62.0] | ||||||||||||
| Molecule Alteration | Missense mutation | p.V35T+p.E40ED+p.V60I+p.K122KE+p.I135V+p.S162A+p.K173T+p.Q174EK+p.P176PS+p.D177E+p.I178M+p.T200A+p.E204EK+p.Q207E+p.R211K+p.V245Q+p.A272P+p.K277R+p.K281R+p.T286A+p.I293V+p.P294T |
|||||||||||
| Experimental Note | Identified from the Human Clinical Data | ||||||||||||
| Experiment for Molecule Alteration |
GeneSeq assay; PhenoSense GT assay | ||||||||||||
| Experiment for Drug Resistance |
Viral load assay | ||||||||||||
| Mechanism Description | Amino acid changes in IN may contribute to drug resistance or sensitivity. | ||||||||||||
| Key Molecule: HIV1 Reverse transcriptase (HIV1 RT) | [3] | ||||||||||||
| Sensitive Disease | Human immunodeficiency virus infection [ICD-11: 1C62.0] | ||||||||||||
| Molecule Alteration | Missense mutation | p.V35M+p.K49KR+p.V60I+p.R83K+p.K122E+p.I135T+p.S162H+p.E248D+p.A272P+p.E297K |
|||||||||||
| Experimental Note | Identified from the Human Clinical Data | ||||||||||||
| Experiment for Molecule Alteration |
GeneSeq assay; PhenoSense GT assay | ||||||||||||
| Experiment for Drug Resistance |
Viral load assay | ||||||||||||
| Mechanism Description | Amino acid changes in IN may contribute to drug resistance or sensitivity. | ||||||||||||
| Key Molecule: HIV1 Reverse transcriptase (HIV1 RT) | [3] | ||||||||||||
| Sensitive Disease | Human immunodeficiency virus infection [ICD-11: 1C62.0] | ||||||||||||
| Molecule Alteration | Missense mutation | p.A98S+p.K122E+p.A158AS+p.I159IV+p.S162SA+p.D177E+p.G196E+p.R211S+p.A272S+p.T286A+p.P294T |
|||||||||||
| Experimental Note | Identified from the Human Clinical Data | ||||||||||||
| Experiment for Molecule Alteration |
GeneSeq assay; PhenoSense GT assay | ||||||||||||
| Experiment for Drug Resistance |
Viral load assay | ||||||||||||
| Mechanism Description | Amino acid changes in IN may contribute to drug resistance or sensitivity. | ||||||||||||
| Key Molecule: HIV1 Reverse transcriptase (HIV1 RT) | [3] | ||||||||||||
| Sensitive Disease | Human immunodeficiency virus infection [ICD-11: 1C62.0] | ||||||||||||
| Molecule Alteration | Missense mutation | p.F171Y+p.D177E+p.I178IMV+p.T200A+p.Q207T+p.R211K+p.V245E+p.A272P+p.K277KR |
|||||||||||
| Experimental Note | Identified from the Human Clinical Data | ||||||||||||
| Experiment for Molecule Alteration |
GeneSeq assay; PhenoSense GT assay | ||||||||||||
| Experiment for Drug Resistance |
Viral load assay | ||||||||||||
| Mechanism Description | Amino acid changes in IN may contribute to drug resistance or sensitivity. | ||||||||||||
| Key Molecule: HIV1 Reverse transcriptase (HIV1 RT) | [3] | ||||||||||||
| Sensitive Disease | Human immunodeficiency virus infection [ICD-11: 1C62.0] | ||||||||||||
| Molecule Alteration | Missense mutation | p.V35I+p.K122E+p.D123N+p.I135L+p.S162C+p.K173N+p.Q174K+p.V179I+p.G196E+p.A272P+p.A288T |
|||||||||||
| Experimental Note | Identified from the Human Clinical Data | ||||||||||||
| Experiment for Molecule Alteration |
GeneSeq assay; PhenoSense GT assay | ||||||||||||
| Experiment for Drug Resistance |
Viral load assay | ||||||||||||
| Mechanism Description | Amino acid changes in IN may contribute to drug resistance or sensitivity. | ||||||||||||
| Key Molecule: HIV1 Reverse transcriptase (HIV1 RT) | [3] | ||||||||||||
| Sensitive Disease | Human immunodeficiency virus infection [ICD-11: 1C62.0] | ||||||||||||
| Molecule Alteration | Missense mutation | p.E6ED+p.V8VI+p.V35I+p.D86DE+p.K122P+p.D123E+p.I135L+p.T139I+p.D177E+p.Q207QK+p.R211RK+p.A272P+p.T286A+p.A288AG+p.I293IV |
|||||||||||
| Experimental Note | Identified from the Human Clinical Data | ||||||||||||
| Experiment for Molecule Alteration |
GeneSeq assay; PhenoSense GT assay | ||||||||||||
| Experiment for Drug Resistance |
Viral load assay | ||||||||||||
| Mechanism Description | Amino acid changes in IN may contribute to drug resistance or sensitivity. | ||||||||||||
| Key Molecule: HIV1 Reverse transcriptase (HIV1 RT) | [3] | ||||||||||||
| Sensitive Disease | Human immunodeficiency virus infection [ICD-11: 1C62.0] | ||||||||||||
| Molecule Alteration | Missense mutation | p.K20KR+p.V60I+p.S68G+p.I178ILM+p.G196E+p.T200TA+p.E248D+p.A272P+p.G273R+p.K275R+p.K281KR+p.V292I+p.I293V |
|||||||||||
| Experimental Note | Identified from the Human Clinical Data | ||||||||||||
| Experiment for Molecule Alteration |
GeneSeq assay; PhenoSense GT assay | ||||||||||||
| Experiment for Drug Resistance |
Viral load assay | ||||||||||||
| Mechanism Description | Amino acid changes in IN may contribute to drug resistance or sensitivity. | ||||||||||||
| Key Molecule: HIV1 Reverse transcriptase (HIV1 RT) | [3] | ||||||||||||
| Sensitive Disease | Human immunodeficiency virus infection [ICD-11: 1C62.0] | ||||||||||||
| Molecule Alteration | Missense mutation | p.V8I+p.K20R+p.T69TA+p.K122KE+p.D123X+p.I178IM+p.R211K+p.V245VI+p.E248ED+p.A272P+p.K277R+p.I293V+p.P294S+p.E297R |
|||||||||||
| Experimental Note | Identified from the Human Clinical Data | ||||||||||||
| Experiment for Molecule Alteration |
GeneSeq assay; PhenoSense GT assay | ||||||||||||
| Experiment for Drug Resistance |
Viral load assay | ||||||||||||
| Mechanism Description | Amino acid changes in IN may contribute to drug resistance or sensitivity. | ||||||||||||
| Key Molecule: HIV1 Reverse transcriptase (HIV1 RT) | [3] | ||||||||||||
| Sensitive Disease | Human immunodeficiency virus infection [ICD-11: 1C62.0] | ||||||||||||
| Molecule Alteration | Missense mutation | p.T39TA+p.T69TA+p.K82KR+p.R83RK+p.S162SN+p.P170PL+p.K173Q+p.I178M+p.T200A+p.Q207E+p.R211K+p.P225PL+p.L228LF+p.A272P+p.K277KR+p.L282LF+p.I293V+p.T296S+p.E297A |
|||||||||||
| Experimental Note | Identified from the Human Clinical Data | ||||||||||||
| Experiment for Molecule Alteration |
GeneSeq assay; PhenoSense GT assay | ||||||||||||
| Experiment for Drug Resistance |
Viral load assay | ||||||||||||
| Mechanism Description | Amino acid changes in IN may contribute to drug resistance or sensitivity. | ||||||||||||
| Key Molecule: HIV1 Reverse transcriptase (HIV1 RT) | [3] | ||||||||||||
| Sensitive Disease | Human immunodeficiency virus infection [ICD-11: 1C62.0] | ||||||||||||
| Molecule Alteration | Missense mutation | p.V35I+p.L80LF+p.A98S+p.K122E+p.T128TA+p.K166KR+p.I178IL+p.R211K+p.V245E+p.K277R+p.I293V+p.P294A+p.E297K |
|||||||||||
| Experimental Note | Identified from the Human Clinical Data | ||||||||||||
| Experiment for Molecule Alteration |
GeneSeq assay; PhenoSense GT assay | ||||||||||||
| Experiment for Drug Resistance |
Viral load assay | ||||||||||||
| Mechanism Description | Amino acid changes in IN may contribute to drug resistance or sensitivity. | ||||||||||||
| Key Molecule: HIV1 Reverse transcriptase (HIV1 RT) | [3] | ||||||||||||
| Sensitive Disease | Human immunodeficiency virus infection [ICD-11: 1C62.0] | ||||||||||||
| Molecule Alteration | Missense mutation | p.K20R+p.V35L+p.T39A+p.R83RK+p.K122E+p.I135R+p.K173E+p.D177E+p.V179I+p.G196E+p.T200I+p.I202V+p.R211K+p.V245VE+p.A288S+p.I293V |
|||||||||||
| Experimental Note | Identified from the Human Clinical Data | ||||||||||||
| Experiment for Molecule Alteration |
GeneSeq assay; PhenoSense GT assay | ||||||||||||
| Experiment for Drug Resistance |
Viral load assay | ||||||||||||
| Mechanism Description | Amino acid changes in IN may contribute to drug resistance or sensitivity. | ||||||||||||
| Key Molecule: HIV1 Reverse transcriptase (HIV1 RT) | [2] | ||||||||||||
| Sensitive Disease | Human immunodeficiency virus infection [ICD-11: 1C62.0] | ||||||||||||
| Molecule Alteration | Missense mutation | p.E6D+p.V106I+p.V118I+p.I135V+p.T165I+p.T200A+p.F214L+p.V245R+p.D250E+p.A272P |
|||||||||||
| Experimental Note | Identified from the Human Clinical Data | ||||||||||||
| Experiment for Molecule Alteration |
Site-directed mutagenesis; Sanger sequencing assay | ||||||||||||
| Experiment for Drug Resistance |
PhenoSense assay | ||||||||||||
| Mechanism Description | Sixteen mutations at eight positions had a mean regression coefficient >=1.0 log10 (a contribution to decreased susceptibility of 10-fold or greater) for nevirapine, efavirenz, etravirine and/or rilpivirine: L100I, k101P, k103N/S, V106A/M, Y181C/I/V, Y188C/L, G190A/E/Q/S and F227C. With the exception of L100I and F227C, each mutation had a mean regression coefficient >=1.0 log10 for nevirapine. With the exception of k103S, V106A, Y181C/I/V and F227C, each had a mean regression coefficient >=1.0 log10 for efavirenz. Five mutations, k101P, Y181I/V, G190E and F227C, had a mean regression coefficient >=1.0 log10 for etravirine. Six mutations, L100I, k101P, Y181I/V, G190E and F227C, had a mean regression coefficient >= 1.0 log10 for rilpivirine. | ||||||||||||
| Key Molecule: HIV1 Reverse transcriptase (HIV1 RT) | [2] | ||||||||||||
| Sensitive Disease | Human immunodeficiency virus infection [ICD-11: 1C62.0] | ||||||||||||
| Molecule Alteration | Missense mutation | p.M41L+p.K43Q+p.E44D+p.V60I+p.D67N+p.G93GR+p.K101H+p.V118I+p.K122P+p.D123E+p.I135T+p.I142V+p.D177E+p.I178IM+p.M184V+p.G190A+p.G196E+p.E203K+p.Q207E+p.L210W+p.R211K+p.T215Y+p.A272P+p.K277R |
|||||||||||
| Experimental Note | Identified from the Human Clinical Data | ||||||||||||
| Experiment for Molecule Alteration |
Site-directed mutagenesis; Sanger sequencing assay | ||||||||||||
| Experiment for Drug Resistance |
PhenoSense assay | ||||||||||||
| Mechanism Description | Sixteen mutations at eight positions had a mean regression coefficient >=1.0 log10 (a contribution to decreased susceptibility of 10-fold or greater) for nevirapine, efavirenz, etravirine and/or rilpivirine: L100I, k101P, k103N/S, V106A/M, Y181C/I/V, Y188C/L, G190A/E/Q/S and F227C. With the exception of L100I and F227C, each mutation had a mean regression coefficient >=1.0 log10 for nevirapine. With the exception of k103S, V106A, Y181C/I/V and F227C, each had a mean regression coefficient >=1.0 log10 for efavirenz. Five mutations, k101P, Y181I/V, G190E and F227C, had a mean regression coefficient >=1.0 log10 for etravirine. Six mutations, L100I, k101P, Y181I/V, G190E and F227C, had a mean regression coefficient >= 1.0 log10 for rilpivirine. | ||||||||||||
| Key Molecule: HIV1 Reverse transcriptase (HIV1 RT) | [2] | ||||||||||||
| Sensitive Disease | Human immunodeficiency virus infection [ICD-11: 1C62.0] | ||||||||||||
| Molecule Alteration | Missense mutation | p.K64KR+p.K102KR+p.D123E+p.K173X+p.P176PAT+p.D177DE+p.I178L+p.M184MV+p.Q197QEK+p.Q207E+p.R211K+p.V245VI+p.E248D+p.A272P+p.E297K |
|||||||||||
| Experimental Note | Identified from the Human Clinical Data | ||||||||||||
| Experiment for Molecule Alteration |
Site-directed mutagenesis; Sanger sequencing assay | ||||||||||||
| Experiment for Drug Resistance |
PhenoSense assay | ||||||||||||
| Mechanism Description | Sixteen mutations at eight positions had a mean regression coefficient >=1.0 log10 (a contribution to decreased susceptibility of 10-fold or greater) for nevirapine, efavirenz, etravirine and/or rilpivirine: L100I, k101P, k103N/S, V106A/M, Y181C/I/V, Y188C/L, G190A/E/Q/S and F227C. With the exception of L100I and F227C, each mutation had a mean regression coefficient >=1.0 log10 for nevirapine. With the exception of k103S, V106A, Y181C/I/V and F227C, each had a mean regression coefficient >=1.0 log10 for efavirenz. Five mutations, k101P, Y181I/V, G190E and F227C, had a mean regression coefficient >=1.0 log10 for etravirine. Six mutations, L100I, k101P, Y181I/V, G190E and F227C, had a mean regression coefficient >= 1.0 log10 for rilpivirine. | ||||||||||||
| Key Molecule: HIV1 Reverse transcriptase (HIV1 RT) | [2] | ||||||||||||
| Sensitive Disease | Human immunodeficiency virus infection [ICD-11: 1C62.0] | ||||||||||||
| Molecule Alteration | Missense mutation | p.V35I+p.S48T+p.K101KR+p.K103N+p.V106VI+p.T139TA+p.P157PS+p.T200I+p.E203D+p.Q207E+p.R211RK+p.D250E+p.A272P+p.K275R+p.K277R+p.T286TA+p.I293IV |
|||||||||||
| Experimental Note | Identified from the Human Clinical Data | ||||||||||||
| Experiment for Molecule Alteration |
Site-directed mutagenesis; Sanger sequencing assay | ||||||||||||
| Experiment for Drug Resistance |
PhenoSense assay | ||||||||||||
| Mechanism Description | Sixteen mutations at eight positions had a mean regression coefficient >=1.0 log10 (a contribution to decreased susceptibility of 10-fold or greater) for nevirapine, efavirenz, etravirine and/or rilpivirine: L100I, k101P, k103N/S, V106A/M, Y181C/I/V, Y188C/L, G190A/E/Q/S and F227C. With the exception of L100I and F227C, each mutation had a mean regression coefficient >=1.0 log10 for nevirapine. With the exception of k103S, V106A, Y181C/I/V and F227C, each had a mean regression coefficient >=1.0 log10 for efavirenz. Five mutations, k101P, Y181I/V, G190E and F227C, had a mean regression coefficient >=1.0 log10 for etravirine. Six mutations, L100I, k101P, Y181I/V, G190E and F227C, had a mean regression coefficient >= 1.0 log10 for rilpivirine. | ||||||||||||
| Key Molecule: HIV1 Reverse transcriptase (HIV1 RT) | [2] | ||||||||||||
| Sensitive Disease | Human immunodeficiency virus infection [ICD-11: 1C62.0] | ||||||||||||
| Molecule Alteration | Missense mutation | p.P4T+p.E6K+p.K20R+p.T39TK+p.M41L+p.K43E+p.E44AD+p.D67N+p.L74I+p.K101E+p.V118I+p.K122E+p.S162A+p.V179F+p.Y181C+p.M184V+p.G190A+p.I195K+p.L210W+p.R211K+p.T215Y+p.K219N+p.V245K+p.I293V+p.E297A |
|||||||||||
| Experimental Note | Identified from the Human Clinical Data | ||||||||||||
| Experiment for Molecule Alteration |
Site-directed mutagenesis; Sanger sequencing assay | ||||||||||||
| Experiment for Drug Resistance |
PhenoSense assay | ||||||||||||
| Mechanism Description | Sixteen mutations at eight positions had a mean regression coefficient >=1.0 log10 (a contribution to decreased susceptibility of 10-fold or greater) for nevirapine, efavirenz, etravirine and/or rilpivirine: L100I, k101P, k103N/S, V106A/M, Y181C/I/V, Y188C/L, G190A/E/Q/S and F227C. With the exception of L100I and F227C, each mutation had a mean regression coefficient >=1.0 log10 for nevirapine. With the exception of k103S, V106A, Y181C/I/V and F227C, each had a mean regression coefficient >=1.0 log10 for efavirenz. Five mutations, k101P, Y181I/V, G190E and F227C, had a mean regression coefficient >=1.0 log10 for etravirine. Six mutations, L100I, k101P, Y181I/V, G190E and F227C, had a mean regression coefficient >= 1.0 log10 for rilpivirine. | ||||||||||||
| Key Molecule: HIV1 Reverse transcriptase (HIV1 RT) | [2] | ||||||||||||
| Sensitive Disease | Human immunodeficiency virus infection [ICD-11: 1C62.0] | ||||||||||||
| Molecule Alteration | Missense mutation | p.K20R+p.K122E+p.R211K+p.V245M+p.G262GE+p.A272P+p.V292VI+p.I293IV+p.E297A |
|||||||||||
| Experimental Note | Identified from the Human Clinical Data | ||||||||||||
| Experiment for Molecule Alteration |
Site-directed mutagenesis; Sanger sequencing assay | ||||||||||||
| Experiment for Drug Resistance |
PhenoSense assay | ||||||||||||
| Mechanism Description | Sixteen mutations at eight positions had a mean regression coefficient >=1.0 log10 (a contribution to decreased susceptibility of 10-fold or greater) for nevirapine, efavirenz, etravirine and/or rilpivirine: L100I, k101P, k103N/S, V106A/M, Y181C/I/V, Y188C/L, G190A/E/Q/S and F227C. With the exception of L100I and F227C, each mutation had a mean regression coefficient >=1.0 log10 for nevirapine. With the exception of k103S, V106A, Y181C/I/V and F227C, each had a mean regression coefficient >=1.0 log10 for efavirenz. Five mutations, k101P, Y181I/V, G190E and F227C, had a mean regression coefficient >=1.0 log10 for etravirine. Six mutations, L100I, k101P, Y181I/V, G190E and F227C, had a mean regression coefficient >= 1.0 log10 for rilpivirine. | ||||||||||||
| Key Molecule: HIV1 Reverse transcriptase (HIV1 RT) | [2] | ||||||||||||
| Sensitive Disease | Human immunodeficiency virus infection [ICD-11: 1C62.0] | ||||||||||||
| Molecule Alteration | Missense mutation | p.P4S+p.E6D+p.K122E+p.D123N+p.I142V+p.S162C+p.D177E+p.I178L+p.R211K+p.A272P+p.I293V+p.E297R |
|||||||||||
| Experimental Note | Identified from the Human Clinical Data | ||||||||||||
| Experiment for Molecule Alteration |
Site-directed mutagenesis; Sanger sequencing assay | ||||||||||||
| Experiment for Drug Resistance |
PhenoSense assay | ||||||||||||
| Mechanism Description | Sixteen mutations at eight positions had a mean regression coefficient >=1.0 log10 (a contribution to decreased susceptibility of 10-fold or greater) for nevirapine, efavirenz, etravirine and/or rilpivirine: L100I, k101P, k103N/S, V106A/M, Y181C/I/V, Y188C/L, G190A/E/Q/S and F227C. With the exception of L100I and F227C, each mutation had a mean regression coefficient >=1.0 log10 for nevirapine. With the exception of k103S, V106A, Y181C/I/V and F227C, each had a mean regression coefficient >=1.0 log10 for efavirenz. Five mutations, k101P, Y181I/V, G190E and F227C, had a mean regression coefficient >=1.0 log10 for etravirine. Six mutations, L100I, k101P, Y181I/V, G190E and F227C, had a mean regression coefficient >= 1.0 log10 for rilpivirine. | ||||||||||||
| Key Molecule: HIV1 Reverse transcriptase (HIV1 RT) | [2] | ||||||||||||
| Sensitive Disease | Human immunodeficiency virus infection [ICD-11: 1C62.0] | ||||||||||||
| Molecule Alteration | Missense mutation | p.E6K+p.V35I+p.T39TI+p.K102KQ+p.V106VI+p.K122E+p.I135IMV+p.I142M+p.S162C+p.R211K+p.K277R+p.A288S+p.I293V |
|||||||||||
| Experimental Note | Identified from the Human Clinical Data | ||||||||||||
| Experiment for Molecule Alteration |
Site-directed mutagenesis; Sanger sequencing assay | ||||||||||||
| Experiment for Drug Resistance |
PhenoSense assay | ||||||||||||
| Mechanism Description | Sixteen mutations at eight positions had a mean regression coefficient >=1.0 log10 (a contribution to decreased susceptibility of 10-fold or greater) for nevirapine, efavirenz, etravirine and/or rilpivirine: L100I, k101P, k103N/S, V106A/M, Y181C/I/V, Y188C/L, G190A/E/Q/S and F227C. With the exception of L100I and F227C, each mutation had a mean regression coefficient >=1.0 log10 for nevirapine. With the exception of k103S, V106A, Y181C/I/V and F227C, each had a mean regression coefficient >=1.0 log10 for efavirenz. Five mutations, k101P, Y181I/V, G190E and F227C, had a mean regression coefficient >=1.0 log10 for etravirine. Six mutations, L100I, k101P, Y181I/V, G190E and F227C, had a mean regression coefficient >= 1.0 log10 for rilpivirine. | ||||||||||||
| Key Molecule: HIV1 Reverse transcriptase (HIV1 RT) | [2] | ||||||||||||
| Sensitive Disease | Human immunodeficiency virus infection [ICD-11: 1C62.0] | ||||||||||||
| Molecule Alteration | Missense mutation | p.K20R+p.I50IV+p.R83K+p.K166KR+p.K173X+p.Q174K+p.D177DE+p.I178L+p.V189VI+p.Q197QK+p.H198GE+p.Q207E+p.R211K+p.V245EQ+p.D250E+p.A272P+p.K277R |
|||||||||||
| Experimental Note | Identified from the Human Clinical Data | ||||||||||||
| Experiment for Molecule Alteration |
Site-directed mutagenesis; Sanger sequencing assay | ||||||||||||
| Experiment for Drug Resistance |
PhenoSense assay | ||||||||||||
| Mechanism Description | Sixteen mutations at eight positions had a mean regression coefficient >=1.0 log10 (a contribution to decreased susceptibility of 10-fold or greater) for nevirapine, efavirenz, etravirine and/or rilpivirine: L100I, k101P, k103N/S, V106A/M, Y181C/I/V, Y188C/L, G190A/E/Q/S and F227C. With the exception of L100I and F227C, each mutation had a mean regression coefficient >=1.0 log10 for nevirapine. With the exception of k103S, V106A, Y181C/I/V and F227C, each had a mean regression coefficient >=1.0 log10 for efavirenz. Five mutations, k101P, Y181I/V, G190E and F227C, had a mean regression coefficient >=1.0 log10 for etravirine. Six mutations, L100I, k101P, Y181I/V, G190E and F227C, had a mean regression coefficient >= 1.0 log10 for rilpivirine. | ||||||||||||
| Key Molecule: HIV1 Reverse transcriptase (HIV1 RT) | [2] | ||||||||||||
| Sensitive Disease | Human immunodeficiency virus infection [ICD-11: 1C62.0] | ||||||||||||
| Molecule Alteration | Missense mutation | p.V35T+p.T39TR+p.K122KE+p.D177E+p.K281R+p.T286A+p.I293IV+p.E297K |
|||||||||||
| Experimental Note | Identified from the Human Clinical Data | ||||||||||||
| Experiment for Molecule Alteration |
Site-directed mutagenesis; Sanger sequencing assay | ||||||||||||
| Experiment for Drug Resistance |
PhenoSense assay | ||||||||||||
| Mechanism Description | Sixteen mutations at eight positions had a mean regression coefficient >=1.0 log10 (a contribution to decreased susceptibility of 10-fold or greater) for nevirapine, efavirenz, etravirine and/or rilpivirine: L100I, k101P, k103N/S, V106A/M, Y181C/I/V, Y188C/L, G190A/E/Q/S and F227C. With the exception of L100I and F227C, each mutation had a mean regression coefficient >=1.0 log10 for nevirapine. With the exception of k103S, V106A, Y181C/I/V and F227C, each had a mean regression coefficient >=1.0 log10 for efavirenz. Five mutations, k101P, Y181I/V, G190E and F227C, had a mean regression coefficient >=1.0 log10 for etravirine. Six mutations, L100I, k101P, Y181I/V, G190E and F227C, had a mean regression coefficient >= 1.0 log10 for rilpivirine. | ||||||||||||
| Key Molecule: HIV1 Reverse transcriptase (HIV1 RT) | [2] | ||||||||||||
| Sensitive Disease | Human immunodeficiency virus infection [ICD-11: 1C62.0] | ||||||||||||
| Molecule Alteration | Missense mutation | p.R83K+p.K101KE+p.K122E+p.D123DE+p.S162A+p.Q174G+p.G196E+p.T200A+p.Q207E+p.V245E+p.A272P+p.K277R+p.I293V |
|||||||||||
| Experimental Note | Identified from the Human Clinical Data | ||||||||||||
| Experiment for Molecule Alteration |
Site-directed mutagenesis; Sanger sequencing assay | ||||||||||||
| Experiment for Drug Resistance |
PhenoSense assay | ||||||||||||
| Mechanism Description | Sixteen mutations at eight positions had a mean regression coefficient >=1.0 log10 (a contribution to decreased susceptibility of 10-fold or greater) for nevirapine, efavirenz, etravirine and/or rilpivirine: L100I, k101P, k103N/S, V106A/M, Y181C/I/V, Y188C/L, G190A/E/Q/S and F227C. With the exception of L100I and F227C, each mutation had a mean regression coefficient >=1.0 log10 for nevirapine. With the exception of k103S, V106A, Y181C/I/V and F227C, each had a mean regression coefficient >=1.0 log10 for efavirenz. Five mutations, k101P, Y181I/V, G190E and F227C, had a mean regression coefficient >=1.0 log10 for etravirine. Six mutations, L100I, k101P, Y181I/V, G190E and F227C, had a mean regression coefficient >= 1.0 log10 for rilpivirine. | ||||||||||||
| Key Molecule: HIV1 Reverse transcriptase (HIV1 RT) | [2] | ||||||||||||
| Sensitive Disease | Human immunodeficiency virus infection [ICD-11: 1C62.0] | ||||||||||||
| Molecule Alteration | Missense mutation | p.V35T+p.T39E+p.K46T+p.E53D+p.V60I+p.K64KR+p.K101Q+p.K104KR+p.K122E+p.D123S+p.I135K+p.K166R+p.K173A+p.D177E+p.T200A+p.Q207E+p.R211K+p.V245E+p.D250E+p.V254I+p.A272P+p.K277R+p.T286A+p.E291D+p.V292I+p.I293V |
|||||||||||
| Experimental Note | Identified from the Human Clinical Data | ||||||||||||
| Experiment for Molecule Alteration |
Site-directed mutagenesis; Sanger sequencing assay | ||||||||||||
| Experiment for Drug Resistance |
PhenoSense assay | ||||||||||||
| Mechanism Description | Sixteen mutations at eight positions had a mean regression coefficient >=1.0 log10 (a contribution to decreased susceptibility of 10-fold or greater) for nevirapine, efavirenz, etravirine and/or rilpivirine: L100I, k101P, k103N/S, V106A/M, Y181C/I/V, Y188C/L, G190A/E/Q/S and F227C. With the exception of L100I and F227C, each mutation had a mean regression coefficient >=1.0 log10 for nevirapine. With the exception of k103S, V106A, Y181C/I/V and F227C, each had a mean regression coefficient >=1.0 log10 for efavirenz. Five mutations, k101P, Y181I/V, G190E and F227C, had a mean regression coefficient >=1.0 log10 for etravirine. Six mutations, L100I, k101P, Y181I/V, G190E and F227C, had a mean regression coefficient >= 1.0 log10 for rilpivirine. | ||||||||||||
| Key Molecule: HIV1 Reverse transcriptase (HIV1 RT) | [2] | ||||||||||||
| Sensitive Disease | Human immunodeficiency virus infection [ICD-11: 1C62.0] | ||||||||||||
| Molecule Alteration | Missense mutation | p.I135V+p.P176S+p.D177E+p.A272P+p.G285A+p.E297K |
|||||||||||
| Experimental Note | Identified from the Human Clinical Data | ||||||||||||
| Experiment for Molecule Alteration |
Site-directed mutagenesis; Sanger sequencing assay | ||||||||||||
| Experiment for Drug Resistance |
PhenoSense assay | ||||||||||||
| Mechanism Description | Sixteen mutations at eight positions had a mean regression coefficient >=1.0 log10 (a contribution to decreased susceptibility of 10-fold or greater) for nevirapine, efavirenz, etravirine and/or rilpivirine: L100I, k101P, k103N/S, V106A/M, Y181C/I/V, Y188C/L, G190A/E/Q/S and F227C. With the exception of L100I and F227C, each mutation had a mean regression coefficient >=1.0 log10 for nevirapine. With the exception of k103S, V106A, Y181C/I/V and F227C, each had a mean regression coefficient >=1.0 log10 for efavirenz. Five mutations, k101P, Y181I/V, G190E and F227C, had a mean regression coefficient >=1.0 log10 for etravirine. Six mutations, L100I, k101P, Y181I/V, G190E and F227C, had a mean regression coefficient >= 1.0 log10 for rilpivirine. | ||||||||||||
| Key Molecule: HIV1 Reverse transcriptase (HIV1 RT) | [2] | ||||||||||||
| Sensitive Disease | Human immunodeficiency virus infection [ICD-11: 1C62.0] | ||||||||||||
| Molecule Alteration | Missense mutation | p.K13KR+p.K122E+p.I135T+p.S162C+p.M184V+p.G196E+p.K277R+p.I293V+p.E297A |
|||||||||||
| Experimental Note | Identified from the Human Clinical Data | ||||||||||||
| Experiment for Molecule Alteration |
Site-directed mutagenesis; Sanger sequencing assay | ||||||||||||
| Experiment for Drug Resistance |
PhenoSense assay | ||||||||||||
| Mechanism Description | Sixteen mutations at eight positions had a mean regression coefficient >=1.0 log10 (a contribution to decreased susceptibility of 10-fold or greater) for nevirapine, efavirenz, etravirine and/or rilpivirine: L100I, k101P, k103N/S, V106A/M, Y181C/I/V, Y188C/L, G190A/E/Q/S and F227C. With the exception of L100I and F227C, each mutation had a mean regression coefficient >=1.0 log10 for nevirapine. With the exception of k103S, V106A, Y181C/I/V and F227C, each had a mean regression coefficient >=1.0 log10 for efavirenz. Five mutations, k101P, Y181I/V, G190E and F227C, had a mean regression coefficient >=1.0 log10 for etravirine. Six mutations, L100I, k101P, Y181I/V, G190E and F227C, had a mean regression coefficient >= 1.0 log10 for rilpivirine. | ||||||||||||
| Key Molecule: HIV1 Reverse transcriptase (HIV1 RT) | [2] | ||||||||||||
| Sensitive Disease | Human immunodeficiency virus infection [ICD-11: 1C62.0] | ||||||||||||
| Molecule Alteration | Missense mutation | p.K20R+p.V35M+p.T69D+p.K102Q+p.K122KE+p.E138EAG+p.S162C+p.I178L+p.M184MV+p.Q207E+p.A272P+p.K277R+p.L283I+p.I293V+p.E297A |
|||||||||||
| Experimental Note | Identified from the Human Clinical Data | ||||||||||||
| Experiment for Molecule Alteration |
Site-directed mutagenesis; Sanger sequencing assay | ||||||||||||
| Experiment for Drug Resistance |
PhenoSense assay | ||||||||||||
| Mechanism Description | Sixteen mutations at eight positions had a mean regression coefficient >=1.0 log10 (a contribution to decreased susceptibility of 10-fold or greater) for nevirapine, efavirenz, etravirine and/or rilpivirine: L100I, k101P, k103N/S, V106A/M, Y181C/I/V, Y188C/L, G190A/E/Q/S and F227C. With the exception of L100I and F227C, each mutation had a mean regression coefficient >=1.0 log10 for nevirapine. With the exception of k103S, V106A, Y181C/I/V and F227C, each had a mean regression coefficient >=1.0 log10 for efavirenz. Five mutations, k101P, Y181I/V, G190E and F227C, had a mean regression coefficient >=1.0 log10 for etravirine. Six mutations, L100I, k101P, Y181I/V, G190E and F227C, had a mean regression coefficient >= 1.0 log10 for rilpivirine. | ||||||||||||
| Key Molecule: HIV1 Reverse transcriptase (HIV1 RT) | [2] | ||||||||||||
| Sensitive Disease | Human immunodeficiency virus infection [ICD-11: 1C62.0] | ||||||||||||
| Molecule Alteration | Missense mutation | p.K20R+p.V60I+p.V90I+p.K101Q+p.K103N+p.V108I+p.I135V+p.I142V+p.D177E+p.M184I+p.T200I+p.R211K+p.T215D+p.A272P+p.V276I+p.L283I+p.I293V+p.E302EG |
|||||||||||
| Experimental Note | Identified from the Human Clinical Data | ||||||||||||
| Experiment for Molecule Alteration |
Site-directed mutagenesis; Sanger sequencing assay | ||||||||||||
| Experiment for Drug Resistance |
PhenoSense assay | ||||||||||||
| Mechanism Description | Sixteen mutations at eight positions had a mean regression coefficient >=1.0 log10 (a contribution to decreased susceptibility of 10-fold or greater) for nevirapine, efavirenz, etravirine and/or rilpivirine: L100I, k101P, k103N/S, V106A/M, Y181C/I/V, Y188C/L, G190A/E/Q/S and F227C. With the exception of L100I and F227C, each mutation had a mean regression coefficient >=1.0 log10 for nevirapine. With the exception of k103S, V106A, Y181C/I/V and F227C, each had a mean regression coefficient >=1.0 log10 for efavirenz. Five mutations, k101P, Y181I/V, G190E and F227C, had a mean regression coefficient >=1.0 log10 for etravirine. Six mutations, L100I, k101P, Y181I/V, G190E and F227C, had a mean regression coefficient >= 1.0 log10 for rilpivirine. | ||||||||||||
| Key Molecule: HIV1 Reverse transcriptase (HIV1 RT) | [2] | ||||||||||||
| Sensitive Disease | Human immunodeficiency virus infection [ICD-11: 1C62.0] | ||||||||||||
| Molecule Alteration | Missense mutation | p.P4S+p.K122KE+p.D123E+p.E169D+p.I178L+p.T200A+p.I202IV+p.R211K+p.L246LP+p.E248D+p.A272P+p.T286TA+p.V292VI+p.I293V |
|||||||||||
| Experimental Note | Identified from the Human Clinical Data | ||||||||||||
| Experiment for Molecule Alteration |
Site-directed mutagenesis; Sanger sequencing assay | ||||||||||||
| Experiment for Drug Resistance |
PhenoSense assay | ||||||||||||
| Mechanism Description | Sixteen mutations at eight positions had a mean regression coefficient >=1.0 log10 (a contribution to decreased susceptibility of 10-fold or greater) for nevirapine, efavirenz, etravirine and/or rilpivirine: L100I, k101P, k103N/S, V106A/M, Y181C/I/V, Y188C/L, G190A/E/Q/S and F227C. With the exception of L100I and F227C, each mutation had a mean regression coefficient >=1.0 log10 for nevirapine. With the exception of k103S, V106A, Y181C/I/V and F227C, each had a mean regression coefficient >=1.0 log10 for efavirenz. Five mutations, k101P, Y181I/V, G190E and F227C, had a mean regression coefficient >=1.0 log10 for etravirine. Six mutations, L100I, k101P, Y181I/V, G190E and F227C, had a mean regression coefficient >= 1.0 log10 for rilpivirine. | ||||||||||||
| Key Molecule: HIV1 Reverse transcriptase (HIV1 RT) | [2] | ||||||||||||
| Sensitive Disease | Human immunodeficiency virus infection [ICD-11: 1C62.0] | ||||||||||||
| Molecule Alteration | Missense mutation | p.V35VIL+p.K103N+p.K122E+p.K173Q+p.I178M+p.M184MV+p.T200TA+p.V245M+p.E248D+p.D250DE+p.T286A+p.A288S |
|||||||||||
| Experimental Note | Identified from the Human Clinical Data | ||||||||||||
| Experiment for Molecule Alteration |
Site-directed mutagenesis; Sanger sequencing assay | ||||||||||||
| Experiment for Drug Resistance |
PhenoSense assay | ||||||||||||
| Mechanism Description | Sixteen mutations at eight positions had a mean regression coefficient >=1.0 log10 (a contribution to decreased susceptibility of 10-fold or greater) for nevirapine, efavirenz, etravirine and/or rilpivirine: L100I, k101P, k103N/S, V106A/M, Y181C/I/V, Y188C/L, G190A/E/Q/S and F227C. With the exception of L100I and F227C, each mutation had a mean regression coefficient >=1.0 log10 for nevirapine. With the exception of k103S, V106A, Y181C/I/V and F227C, each had a mean regression coefficient >=1.0 log10 for efavirenz. Five mutations, k101P, Y181I/V, G190E and F227C, had a mean regression coefficient >=1.0 log10 for etravirine. Six mutations, L100I, k101P, Y181I/V, G190E and F227C, had a mean regression coefficient >= 1.0 log10 for rilpivirine. | ||||||||||||
| Key Molecule: HIV1 Reverse transcriptase (HIV1 RT) | [2] | ||||||||||||
| Sensitive Disease | Human immunodeficiency virus infection [ICD-11: 1C62.0] | ||||||||||||
| Molecule Alteration | Missense mutation | p.V60I+p.I135V+p.D177E+p.R211K+p.I244V+p.V245I+p.D250E+p.A272P+p.K277KR+p.E297V |
|||||||||||
| Experimental Note | Identified from the Human Clinical Data | ||||||||||||
| Experiment for Molecule Alteration |
Site-directed mutagenesis; Sanger sequencing assay | ||||||||||||
| Experiment for Drug Resistance |
PhenoSense assay | ||||||||||||
| Mechanism Description | Sixteen mutations at eight positions had a mean regression coefficient >=1.0 log10 (a contribution to decreased susceptibility of 10-fold or greater) for nevirapine, efavirenz, etravirine and/or rilpivirine: L100I, k101P, k103N/S, V106A/M, Y181C/I/V, Y188C/L, G190A/E/Q/S and F227C. With the exception of L100I and F227C, each mutation had a mean regression coefficient >=1.0 log10 for nevirapine. With the exception of k103S, V106A, Y181C/I/V and F227C, each had a mean regression coefficient >=1.0 log10 for efavirenz. Five mutations, k101P, Y181I/V, G190E and F227C, had a mean regression coefficient >=1.0 log10 for etravirine. Six mutations, L100I, k101P, Y181I/V, G190E and F227C, had a mean regression coefficient >= 1.0 log10 for rilpivirine. | ||||||||||||
| Key Molecule: HIV1 Reverse transcriptase (HIV1 RT) | [2] | ||||||||||||
| Sensitive Disease | Human immunodeficiency virus infection [ICD-11: 1C62.0] | ||||||||||||
| Molecule Alteration | Missense mutation | p.V35I+p.R83RK+p.T165TI+p.F171Y+p.T200A+p.I202V+p.V245M+p.A272P+p.K277R+p.T286A+p.P294Q+p.E297A |
|||||||||||
| Experimental Note | Identified from the Human Clinical Data | ||||||||||||
| Experiment for Molecule Alteration |
Site-directed mutagenesis; Sanger sequencing assay | ||||||||||||
| Experiment for Drug Resistance |
PhenoSense assay | ||||||||||||
| Mechanism Description | Sixteen mutations at eight positions had a mean regression coefficient >=1.0 log10 (a contribution to decreased susceptibility of 10-fold or greater) for nevirapine, efavirenz, etravirine and/or rilpivirine: L100I, k101P, k103N/S, V106A/M, Y181C/I/V, Y188C/L, G190A/E/Q/S and F227C. With the exception of L100I and F227C, each mutation had a mean regression coefficient >=1.0 log10 for nevirapine. With the exception of k103S, V106A, Y181C/I/V and F227C, each had a mean regression coefficient >=1.0 log10 for efavirenz. Five mutations, k101P, Y181I/V, G190E and F227C, had a mean regression coefficient >=1.0 log10 for etravirine. Six mutations, L100I, k101P, Y181I/V, G190E and F227C, had a mean regression coefficient >= 1.0 log10 for rilpivirine. | ||||||||||||
| Key Molecule: HIV1 Reverse transcriptase (HIV1 RT) | [2] | ||||||||||||
| Sensitive Disease | Human immunodeficiency virus infection [ICD-11: 1C62.0] | ||||||||||||
| Molecule Alteration | Missense mutation | p.I135V+p.Y181C+p.M184V+p.G196E+p.T200A+p.Q207E+p.R211K+p.T215Y+p.V245K+p.K277R+p.E291D+p.E297K |
|||||||||||
| Experimental Note | Identified from the Human Clinical Data | ||||||||||||
| Experiment for Molecule Alteration |
Site-directed mutagenesis; Sanger sequencing assay | ||||||||||||
| Experiment for Drug Resistance |
PhenoSense assay | ||||||||||||
| Mechanism Description | Sixteen mutations at eight positions had a mean regression coefficient >=1.0 log10 (a contribution to decreased susceptibility of 10-fold or greater) for nevirapine, efavirenz, etravirine and/or rilpivirine: L100I, k101P, k103N/S, V106A/M, Y181C/I/V, Y188C/L, G190A/E/Q/S and F227C. With the exception of L100I and F227C, each mutation had a mean regression coefficient >=1.0 log10 for nevirapine. With the exception of k103S, V106A, Y181C/I/V and F227C, each had a mean regression coefficient >=1.0 log10 for efavirenz. Five mutations, k101P, Y181I/V, G190E and F227C, had a mean regression coefficient >=1.0 log10 for etravirine. Six mutations, L100I, k101P, Y181I/V, G190E and F227C, had a mean regression coefficient >= 1.0 log10 for rilpivirine. | ||||||||||||
| Key Molecule: HIV1 Reverse transcriptase (HIV1 RT) | [2] | ||||||||||||
| Sensitive Disease | Human immunodeficiency virus infection [ICD-11: 1C62.0] | ||||||||||||
| Molecule Alteration | Missense mutation | p.K20KR+p.T27TS+p.V60I+p.R83K+p.K122KE+p.Q174L+p.I178IL+p.T200X+p.R211RK+p.V245E+p.L283LI+p.T286TA+p.I293V |
|||||||||||
| Experimental Note | Identified from the Human Clinical Data | ||||||||||||
| Experiment for Molecule Alteration |
Site-directed mutagenesis; Sanger sequencing assay | ||||||||||||
| Experiment for Drug Resistance |
PhenoSense assay | ||||||||||||
| Mechanism Description | Sixteen mutations at eight positions had a mean regression coefficient >=1.0 log10 (a contribution to decreased susceptibility of 10-fold or greater) for nevirapine, efavirenz, etravirine and/or rilpivirine: L100I, k101P, k103N/S, V106A/M, Y181C/I/V, Y188C/L, G190A/E/Q/S and F227C. With the exception of L100I and F227C, each mutation had a mean regression coefficient >=1.0 log10 for nevirapine. With the exception of k103S, V106A, Y181C/I/V and F227C, each had a mean regression coefficient >=1.0 log10 for efavirenz. Five mutations, k101P, Y181I/V, G190E and F227C, had a mean regression coefficient >=1.0 log10 for etravirine. Six mutations, L100I, k101P, Y181I/V, G190E and F227C, had a mean regression coefficient >= 1.0 log10 for rilpivirine. | ||||||||||||
References
If you find any error in data or bug in web service, please kindly report it to Dr. Sun and Dr. Yu.
